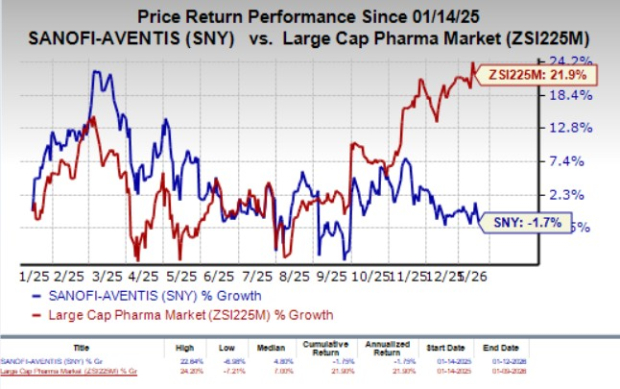
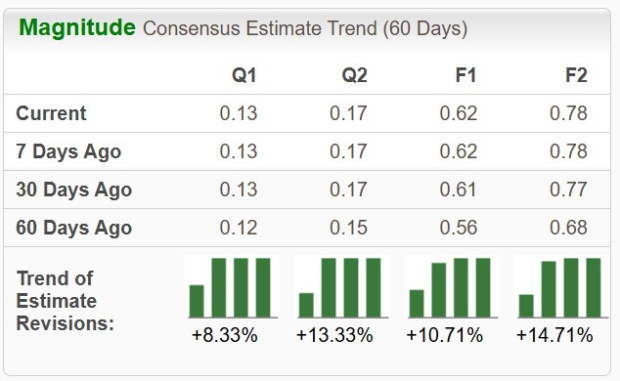
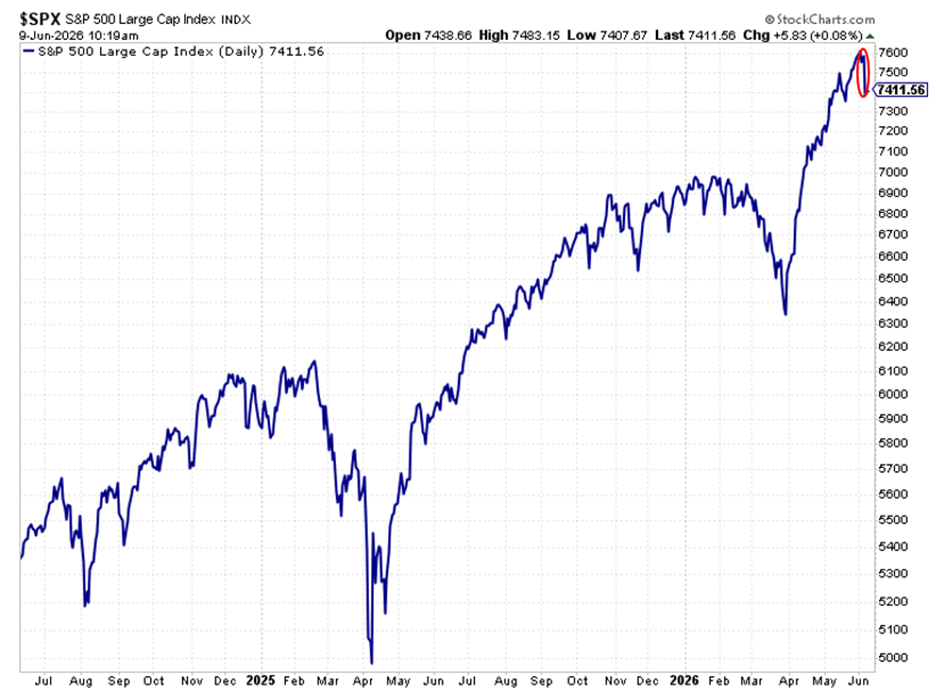
Sanofi (SNY) announced that the European Commission has approved Teizeild (teplizumab) for delaying the onset of stage 3 type 1 diabetes (T1D) in patients aged eight and older with stage 2 disease. This marks Teizeild as the first disease-modifying drug for T1D authorized in the European Union. The drug’s approval follows a positive opinion from the European Medicines Agency in November 2023.
The approval is based on results from the TN-10 phase II study, which involved 76 participants aged 8 to 45. The study demonstrated that participants treated with Teizeild had a median progression time to stage 3 diabetes of 48.4 months, compared to 24.4 months for the placebo group, with 57% of Teizeild patients remaining in stage 2 versus 28% for placebo. Teizeild is already approved in the United States for the same indication, and Sanofi recorded global sales of €47 million for Teizeild in the first nine months of 2025, reflecting a 33.3% year-over-year increase.
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.