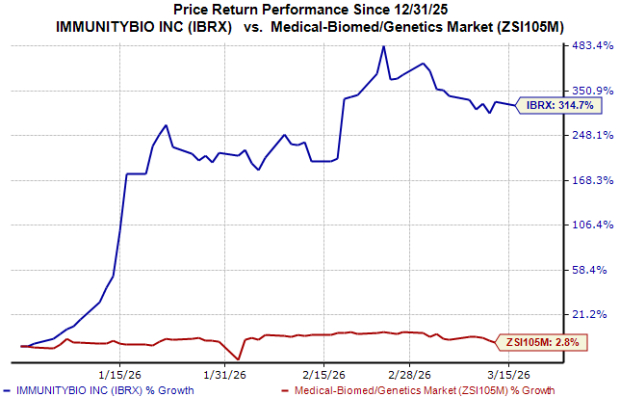
ImmunityBio (IBRX) reported a substantial increase in net product revenues for 2025, totaling $113 million, representing a 700% growth year-over-year. This surge was primarily driven by the strong performance of their FDA-approved product, Anktiva, which is used in conjunction with Bacillus Calmette-Guérin (BCG) to treat adult patients with BCG-unresponsive non-muscle invasive bladder cancer. The ongoing BCG shortage has led to increased demand for Anktiva, and the company expects this momentum to continue.
In addition, IBRX has secured regulatory approvals for Anktiva in the European Union and Saudi Arabia for the same indication in 2026, marking crucial steps in expanding its international market presence. Despite these advancements, ImmunityBio encounters significant competition from established immunotherapy options such as Merck’s Keytruda and Bristol Myers Squibb’s Opdivo, which may hinder its market penetration efforts.