An Industry Milestone
Merck, a stalwart in the pharmaceutical industry, has recently received the FDA’s nod for its groundbreaking activin signaling inhibitor, sotatercept, under the brand name Winrevair. This approval is a much-awaited milestone in the treatment of pulmonary arterial hypertension (PAH), a rare and progressive blood vessel disorder affecting the lungs.
Winrevair’s Triumph in the STELLAR Study
The green light from the FDA was based on the compelling data from the STELLAR study, where Winrevair, in combination with standard background therapy, showcased remarkable outcomes. Notably, the drug significantly enhanced exercise capacity by boosting the six-minute walk distance by 41 meters from the baseline at week 24, meeting the study’s primary objective with flying colors.
This innovation also displayed statistically significant enhancements across eight out of nine secondary measures, demonstrating a reduction in the risk of clinical deterioration or mortality. Winrevair’s safety profile highlights potential increases in hemoglobin levels and the risk of erythrocytosis, a condition marked by excess red blood cells.
Market Response and Future Prospects
Investors heaved a collective sigh of relief as Merck’s stock soared nearly 5% in after-hours trading post the approval of Winrevair. The drug emerges as a cornerstone of Merck’s strategic growth approach, with analysts foreseeing blockbuster potential in the PAH space.
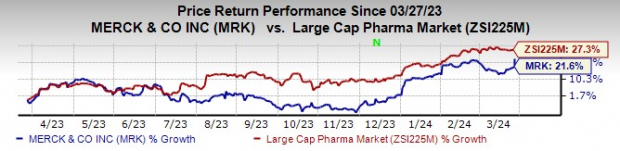
Looking back, Merck’s stock has seen a notable uptick of 21.6% over the past year, albeit lagging slightly behind the industry’s 27.3% rise.
A Breath of Fresh Air in PAH Treatment
PAH exerts a formidable toll on patients as it imposes excessive strain on the heart due to heightened lung blood pressure, potentially leading to heart failure if untreated. Winrevair’s unique mechanism of action targets the intricate cellular signaling pathways associated with vascular hyperproliferation and pathological remodeling, offering a beacon of hope to those grappling with this debilitating condition.
Looking Ahead
Winrevair’s pioneering status as the first novel activin signaling inhibitor endorsed by the FDA for PAH marks a significant leap in the therapeutic landscape. Noteworthy is the FDA’s earlier designations of breakthrough therapy and orphan drug for Winrevair in PAH treatment, reflecting its pivotal role in addressing unmet medical needs.
Merck’s ongoing assessment of sotatercept in more advanced PAH stages through the ZENITH and HYPERION phase III studies underscores its unwavering commitment to pushing the boundaries of medical innovation.
Market Dynamics and Competitors
Key industry players such as United Therapeutics and Johnson & Johnson have also made substantial strides in the PAH domain. United Therapeutics boasts a robust portfolio of four drugs for PAH management in the United States, with Remodulin, Orenitram, Tyvaso, and Adcirca leading the charge.
On the other hand, J&J’s foray into the PAH arena gained traction following its acquisition of Actelion in 2017, propelling its PAH revenue to $3.8 billion in 2023, marking a solid 11.6% year-over-year increase.
Conclusion
As Merck basks in the glory of Winrevair’s approval, the pharmaceutical landscape witnesses a transformative moment in the battle against PAH. With promising advancements and a steadfast commitment to innovation, Merck is poised to redefine the treatment paradigm for patients grappling with PAH’s relentless grip.
Amidst a backdrop of fierce competition and relentless scientific endeavor, the future shines bright for Merck and the broader PAH treatment market, promising a breath of fresh air for all stakeholders involved in this critical healthcare domain.