Ron Finklestien
Eni Reports Strong Q1 Financial Performance but Reduces 2025 Capital Expenditure Forecast
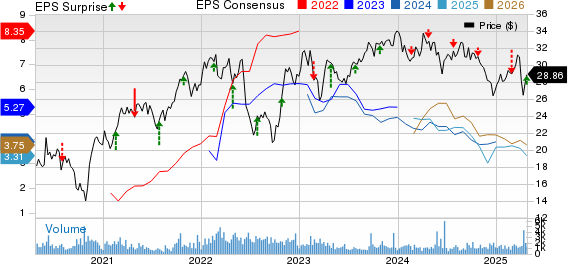
Eni S.p.A Reports Q1 2025 Earnings: Earnings Beat Consensus but Revenues Decline Eni S.p.A reported adjusted earnings of 92 cents per American Depository Receipt ...
Essential Insights Beyond the Hype: Understanding Microsoft Corporation’s (MSFT) Stock Performance
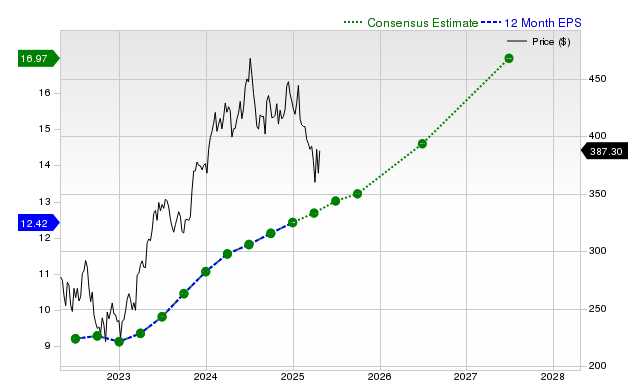
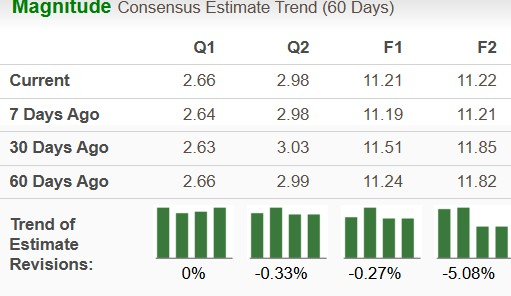
Microsoft’s Stock Performance: Key Metrics and Future Outlook Microsoft (MSFT) has been a focal point in recent stock searches. Several key factors may influence ...
Analyzing GM’s Q1 Earnings: Is Now the Right Time to Invest?
General Motors Set to Report Q1 2025 Results on April 29 General Motors (GM) is expected to announce its first-quarter 2025 results on April ...
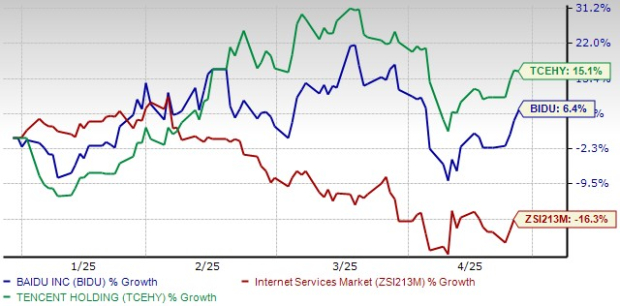
Baidu vs. Tencent: Analyzing the Top Chinese AI Investments for Today’s Market
Comparing Baidu and Tencent: Key Players in AI Growth China’s tech giants Baidu (BIDU) and Tencent Holdings Limited (TCEHY) are significant contenders in the ...
Top Momentum Stock Picks for April 25th
Three Strong Buy Stocks with Momentum for Investors Investors seeking new opportunities may want to consider three stocks that currently hold a buy rating ...
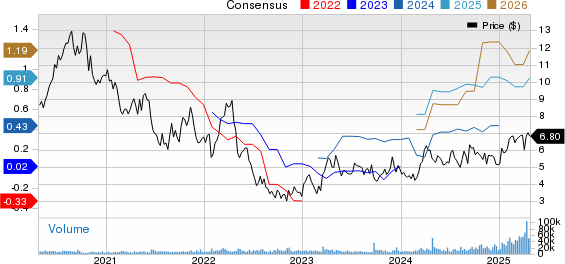
“Understanding Tesla’s 30% Stock Decline: Key Insights Before Investing”
Tesla Stock Faces 30% Decline Amid Market Correction Tesla’s (NASDAQ: TSLA) stock has significantly declined since the start of the year. Currently, shares are ...