Phase III Success for AXS-12
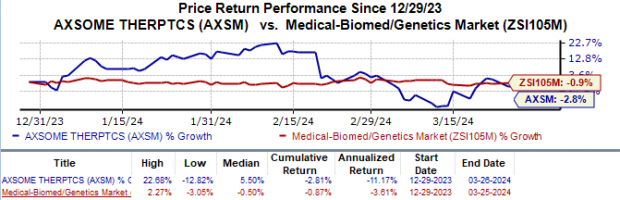
The recently announced results of Axsome Therapeutics’ phase III SYMPHONY study on the investigational narcolepsy drug AXS-12 have sparked optimism in the market. The study’s primary goal of achieving a rapid and significant reduction in weekly cataplexy events was met with flying colors. Impressively, treatment with AXS-12 showcased an 83% decrease in cataplexy attacks over five weeks, outperforming the placebo’s rate of 66%.
Furthermore, the study revealed that AXS-12 achieved a remarkable 56% reduction in cataplexy attacks in the first week alone, compared to the placebo’s 31%. Treatment with AXS-12 also led to an upsurge in remission of cataplexy, with 33% of patients experiencing remission compared to just 9.5% in the placebo group.
Enhanced Tolerance and Benefits
Notably, participants in the study reported a positive tolerability to AXS-12, with additional benefits extending beyond cataplexy reduction. Not only did the drug reduce symptoms like excessive daytime sleepiness (EDS) and unplanned naps but also displayed improvements in cognitive function and memory.
Awaiting the completion of the ongoing open-label extension study to evaluate long-term safety and effectiveness, Axsome is gearing up to present detailed findings from the SYMPHONY study at upcoming medical conferences.
Strategic Acquisition and Market Impact
In a strategic move to diversify its sleep disorder portfolio, Axsome acquired the U.S. rights to Sunosi, a narcolepsy treatment, from Jazz Pharmaceuticals in 2022. With Sunosi becoming a significant revenue source post-acquisition, Axsome’s move demonstrates a keen eye for expanding its market presence.
In a symbiotic relationship, Jazz Pharmaceuticals, known for its sleep disorder drugs Xywav and Xyrem, anticipates substantial revenues from the combined sales of $2 billion by 2025. This synergy between Axsome and Jazz bodes well for mutual growth and innovation in the sleep disorder treatment space.
Market Outlook and Exciting Prospects
Axsome’s narcolepsy drug, AXS-12, harnessing the power of orphan drug designation (ODD) by the FDA, is poised to make a significant impact in the treatment landscape for this chronic sleep disorder. With narcolepsy affecting a substantial portion of the population, the success of AXS-12 holds promise for addressing the unmet needs of patients suffering from this condition.
As Axsome continues to chart new territories with its innovative therapies, investors are keeping a close watch on the company’s growth trajectory. With Sunosi already contributing significantly to Axsome’s revenue stream, the future looks bright for this dynamic player in the pharmaceutical industry.
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.