Bayer Secures Full FDA Approval for Vitrakvi Cancer Treatment
Bayer (BAYRY) has achieved full approval from the FDA for its oncology drug, Vitrakvi (larotrectinib), marking a significant milestone as a first-in-class TRK inhibitor.
The FDA has now fully approved Vitrakvi in the United States for both adult and pediatric patients with solid tumors associated with neurotrophic receptor tyrosine kinase (NTRK) gene fusions. This approval applies specifically to patients without a known acquired resistance mutation who have either had their cancer spread or are not eligible for surgery. Vitrakvi is intended for patients lacking satisfactory alternative treatment options or those whose disease has progressed after previous treatments.
It’s noteworthy that Vitrakvi initially received accelerated approval from the FDA back in November 2018.
Details on Vitrakvi’s Approval
The full FDA approval follows the observation of positive results from confirmatory trials, suggesting Vitrakvi could serve as a new standard of care for treating NTRK gene fusion-positive tumors in both adult and pediatric patients.
This oral TRK inhibitor was specifically designed to target and inhibit the TRK protein family (TRKA, TRKB, and TRKC).
Overall efficacy results from three key multicenter, open-label, single-arm clinical trials—LOXO-TRK-14001 (NCT02122913), SCOUT (NCT02637687), and NAVIGATE (NCT02576431)—indicated a 60% overall response rate, with 24% of patients achieving a complete response and 36% recording a partial response.
Following the acquisition of Loxo Oncology by Eli Lilly and Company in February 2019, Bayer secured exclusive rights for the global development and commercialization of Vitrakvi.
Bayer’s Strategic Focus Moving Forward
As Bayer navigates the upcoming challenges in 2025, it anticipates a decline in earnings coupled with modest sales growth. Uncertainties surrounding ongoing Roundup litigation continue to weigh heavily on the stock’s performance.
In 2024, sales growth in the Pharmaceutical division showed promise, with new products Nubeqa and Kerendia maintaining their momentum. This success has helped counters the downturn attributed to declining sales of Xarelto. The company is actively working to expand the indications for these key drugs to drive future growth.
For Bayer, the successful launch of additional drugs is crucial amid various market challenges. The company is poised to introduce two new treatments: elinzanetant, aimed at alleviating menopause symptoms, and acoramidis, designed for specific heart disease treatment.
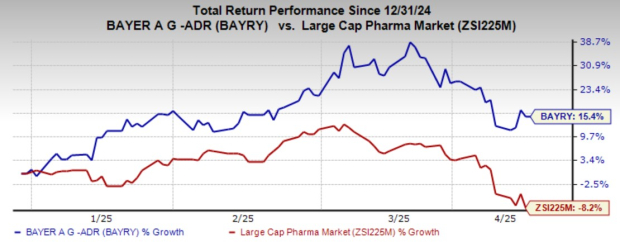
Year-to-date, shares of BAYRY have increased 15.4%, outperforming the industry, which has seen a decline of 8.2%.
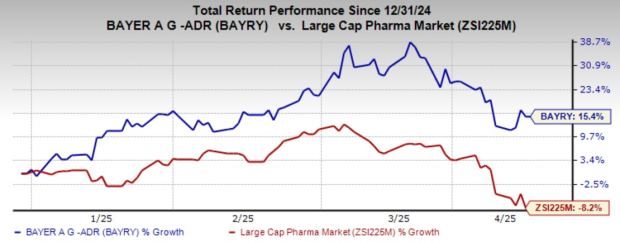
Image Source: Zacks Investment Research
Additionally, Bayer aims to fortify its pharmaceutical pipeline. In 2021, it acquired Vividion Therapeutics, a clinical-stage biopharmaceutical company, to enhance its focus on precision small-molecule therapeutics, particularly in oncology and immunology.
The company has also broadened its pipeline to include new cell therapy avenues through the acquisition of BlueRock and gene therapy via AskBio.
Recently, Bayer announced a licensing agreement with Suzhou Puhe BioPharma Co., which specializes in the research and development of innovative small-molecule precision therapeutics.
Bayer’s Zacks Rank and Recommendations
Currently, BAYRY holds a Zacks Rank #2 (Buy).
Other top-ranked stocks in the pharma and biotech sectors include Amicus Therapeutics, Inc. (FOLD) and ANI Pharmaceuticals, Inc. (ANIP), both rated Zacks Rank #1 (Strong Buy).
Reviewing Amicus Therapeutics
In the past 60 days, earnings per share (EPS) estimates for Amicus have risen from 43 cents to 52 cents for 2025. Additionally, 2026 EPS estimates increased from 71 cents to 79 cents. FOLD has beaten earnings estimates in three of its last four quarters, with an average surprise of 45.42%.
Overview of ANI Pharmaceuticals
Similarly, ANI Pharmaceuticals saw its EPS estimates increase from $5.54 to $6.34 for 2025 over the past two months. For 2026, estimates edged up from $6.90 to $7.13. Year-to-date, ANIP shares have increased by 14%, with earnings surpassing estimates in all four recent quarters, and an average surprise of 17.32%.
Zacks Highlights a Top Semiconductor Stock
Amidst a booming semiconductor market, predicted to expand to $803 billion by 2028, Zacks has identified a top semiconductor stock with potential for significant growth. This company, much smaller than NVIDIA— which has surged over 800% since its recommendation— is well-positioned to meet the growing demand for technologies like Artificial Intelligence and the Internet of Things.
See This Stock Now for Free >>
Bayer Aktiengesellschaft (BAYRY): Free Stock Analysis Report
Amicus Therapeutics, Inc. (FOLD): Free Stock Analysis Report
ANI Pharmaceuticals, Inc. (ANIP): Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
The views and opinions expressed herein are those of the author and do not necessarily reflect those of Nasdaq, Inc.