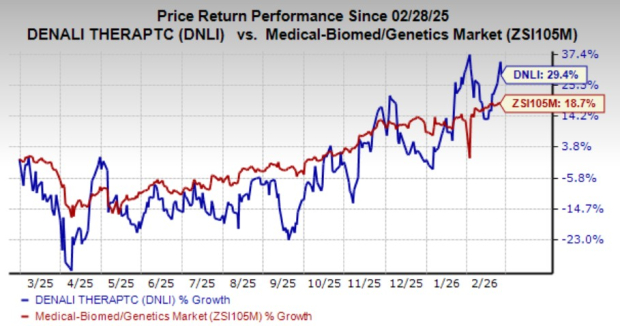
**Denali Therapeutics Reports Q4 2025 Loss**
Denali Therapeutics (DNLI) posted a fourth-quarter loss of 73 cents per share on February 12, 2026, slightly better than the Zacks Consensus Estimate of a 75-cent loss. The company reported a 67-cent loss per share in the same quarter last year. As of December 31, 2025, Denali’s cash and cash equivalents stood at approximately $966.2 million. Notably, the company did not generate any collaboration revenues during the quarter, despite an expected revenue estimate of $18 million.
**Financial Overview and Pipeline Updates**
Total operating expenses rose, leading to a year-over-year increase in losses, with research and development spending declining by 1.9% to $97.9 million, while administrative expenses surged by 31.3% to $39.5 million. Denali is awaiting FDA approval for its lead candidate, tividenofusp alfa, which aims to treat mucopolysaccharidosis type II. The FDA extended the review timeline to April 5, 2026, following Denali’s submission of updated clinical data.
**Future Outlook**
Looking forward, Denali’s pipeline includes DNL126 for Sanfilippo syndrome type A, which recently showed promising preliminary results. The success of tividenofusp alfa and other ongoing studies will significantly influence Denali’s growth prospects, particularly in the face of its substantial cash reserves. The company recorded a total loss per share of $2.97 in 2025, wider than $2.57 in 2024.