FDA Approves Resmetirom for NASH with Liver Scarring
In a groundbreaking move, the FDA has given its conditional nod to Madrigal Pharmaceuticals Inc.’s pioneering drug, Rezdiffra (resmetirom), as a treatment for noncirrhotic nonalcoholic steatohepatitis (NASH) with moderate to advanced liver scarring (fibrosis) – a milestone achievement in the field of liver disease therapeutics. This spectacular development comes with the stipulation that ongoing confirmatory trials corroborate the drug’s clinical benefits to secure continued approval.
Leveraging Advanced Science Against Liver Scarring
Resmetirom, a once-daily oral THR-β agonist, operates at the forefront of medical innovation by targeting the fundamental drivers of NASH. Its accelerated approval was chiefly based on the resounding success of the Phase 3 MAESTRO-NASH trial, where 1,759 patients were enlisted to gauge the drug’s efficacy.
Unprecedented Success in Clinical Trials
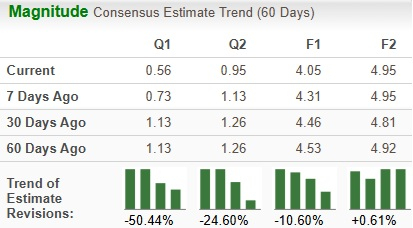
After 52 weeks on treatment, both 100 mg and 80 mg doses of Rezdiffra exhibited statistically significant strides compared to a placebo in achieving two primary endpoints – NASH resolution with no fibrosis deterioration and an improvement in fibrosis by at least one stage without aggravating the NAFLD activity score. To elaborate, liver biopsies indicated that between 24% to 36% of subjects on the 100 mg dosage and 26% to 27% on the 80 mg dose experienced NASH resolution and stable fibrosis, a substantial leap from the 9%-13% observed in the placebo group.
Moreover, a notable 24%-28% of patients on the 100 mg dosage and 23% of those on the 80 mg regimen saw an amelioration in liver scarring alongside no exacerbation of NASH, contrasting sharply with the 13%-15% demographics under placebo treatment.
Paving the Way for a Healthcare Revolution
The approval of Rezdiffra without a mandatory liver biopsy for diagnosis reinforces its accessibility and patient-friendly approach. The ongoing MAESTRO-NASH study and another parallel analysis focusing on liver decompensation events in individuals with well-compensated NASH cirrhosis treated with Rezdiffra signify the drug’s multifaceted potential in addressing various facets of this critical health issue. The drug’s imminent availability in the U.S. slated for April through a specialized pharmacy network heralds a new era for the approximately 6-8 million Americans grappling with NASH and liver scarring.
Market Response and Future Outlook
The approval of Rezdiffra has sparked a significant upward trend for Madrigal Pharmaceuticals, with their shares climbing by 10.67% to $269.55 – a tangible reflection of the investor community’s optimism and the broader market’s recognition of the drug’s potential impact.
Photo: Julia Koblitz via Unsplash
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.