The Power of GCase
Discovering the overlooked potential of Gain Therapeutics (NASDAQ:GANX), the emergence of its drug candidate GT-02287, and recent AI-based partnerships and acquisitions have sparked a renewed interest. GT-02287’s focus on GCase and lysosomal dysfunction intertwines with recent scientific findings, surpassing the potential of Ambroxol, currently in Phase 3 trials for PD.
Gain’s strategic focus on the PD-GBA1 subgroup aligns with its drug’s MoA, setting the stage for a promising future. Despite a temporary stock dip due to recent financing, Gain’s market performance has rebounded, underlining investor confidence. The recent $610 million acquisition of Caraway Therapeutics further solidifies Gain’s approach, positioning it as a more evolved and undervalued alternative.
The Rise of Gain Therapeutics
Introducing Gain Therapeutics
Gain Therapeutics, a biotech firm, utilizes an AI-supported 3D structural biology and physics-based models for drug target discovery. Its early-stage pipeline, including a Phase 1 trial, showcases the potential of its AI-driven drug discovery engine.
The company’s lead product, GT-02287, targets GCase, the primary genetic risk factor for Parkinson’s disease [PD]. Gain is currently enrolling patients in its Phase 1 trial for GBA-1 PD, with data expected in the second half of 2024.
Unlocking Novel Drug Targets
Gain’s innovative drug discovery platform identifies allosteric binding sites, expanding the possibilities within drug targeting. This breakthrough capability breathes new life into proteins previously deemed “undruggable.”

The platform’s unprecedented speed in identifying novel binding sites and the systematic discovery of previously inaccessible targets outshine traditional drug discovery approaches. Gain’s remarkable efficacy and potential industry value warrant further exploration, especially in light of GT-02287’s promising developments.
GT-02287: Unraveling the Potential
The Breakthrough Candidate
GT-02287, Gain’s orally administrable drug candidate, focuses on GCase to combat PD with a GBA-1 mutation and Gaucher disease. GCase’s role in lysosomal function presents a compelling target in the treatment of neurodegenerative diseases.
Unveiling GCase Dysfunction in PD
PD’s prevalence and the intricate relationship between GBA mutations and GCase dysfunction underscore GT-02287’s significance. With a focus on GCase dysfunction and its impact on immune cells and neuroinflammation, GT-02287’s potential to address the unique needs of PD patients is unprecedented.
Revolutionary Developments in the Fight Against Parkinson’s Disease
A Glimpse into the World of GCase and α-synuclein Pathology
The interplay between GCase and α-synuclein in Parkinson’s Disease (PD) has become a focal point of groundbreaking research. Through a bidirectional loop, heightened α-synuclein levels lead to reduced GCase activity, while lower GCase activity results in an increase in α-synuclein. Gain’s rival, Vanqua Bio, has effectively showcased GCase’s pivotal role in the disease.
Ambroxol as a Game-Changing GCase Modulator for PD
Amidst the quest for a PD breakthrough, Ambroxol, a key ingredient in cough syrup, has emerged as a frontrunner in scientific circles. Garnering endorsement from the Michael J. Fox Foundation, Ambroxol is currently undergoing Phase 3 trials for PD. Studies have demonstrated that Ambroxol treatment elevates GCase levels and activity while reducing α-synuclein levels, positioning it as a potential game-changer in the fight against PD.
Unveiling the Dysfunctional GCase Buildup
Recent research has shed light on the accumulation of dysfunctional GCase outside of the lysosome, hinting that simply upregulating the expression of misfolded GCase may not be the panacea for addressing mutant GCase concerns. Mutant GCase has been found to impede alpha-synuclein degradation and aggravate mitochondrial cell metabolism reduction in individuals with GBA1 mutations, aligning with the mitochondrial hypothesis of Parkinson’s. These revelations present a paradigm shift, bringing into question the conventional approach to lysosomal intervention. Essentially, they cast doubt on the effectiveness of merely upregulating GCase without rectifying its misfolded state.
Implications of GCase Dysfunction
The ramifications of GCase dysfunction extend beyond a mere disruption of protein trafficking into the lysosome. They encompass a spectrum of consequences, including the impediment of α-synuclein clearance, mitochondrial dysfunction, impaired glial function and neuroinflammation, ultimately culminating in cellular demise.
Ambroxol and VQ-1010, another promising drug candidate from Vanqua Bio, are envisioned to upregulate GCase levels without rectifying its misfolded state. Consequently, one cannot discount the potential perpetuation of GCase misfolding, hindering its assimilation into the lysosome and impeding the trafficking of other proteins.
The Role of GT-02287 in Addressing GCase Misfolding
GT-02287 assumes a pivotal role in tackling GCase misfolding. By rectifying misfolded GCase within the endoplasmic reticulum (ER), GT-02287 aims to facilitate the entry of normal GCase levels into the lysosome, unleashing their full enzymatic potential. Such a strategy promises a multi-faceted approach, ranging from mitigating α-synuclein levels and enhancing lysosomal function to ameliorating energy metabolism and mitochondrial dysfunction, curbing neuroinflammation, and safeguarding against cellular degeneration.
Efficacy of GT-02287: A Ray of Hope
Gain’s preclinical research endeavors have yielded a wealth of promising results. Notably, these encompass a substantial reduction in alpha-synuclein levels and ameliorated inflammation, evidenced by diminished microgliosis. These hallmark achievements bear paramount significance in the context of combatting Parkinson’s. Furthermore, research highlights GT-02287’s statistically significant therapeutic impact on neurite network and overall lysosomal health in rat neurons. Additionally, in a standard PD model in mice, GT-02287 showcased dose-dependent enhancements in motor deficits, marking a significant milestone in the quest for a panacea. Augmenting its laurels, preclinical data unveiled a remarkable reduction in plasma NfL levels following a 14-day treatment, underscoring the drug’s unwavering potential as a game-changer in the field. NfL, a revered biomarker in both the scientific realm and the FDA domain, adds veracity to these findings, cementing GT-02287’s status as an agent of transformative change in the fight against PD.
Groundbreaking Developments in Neurodegenerative Disease Research
As Gain Therapeutics continues to make significant strides in the field of neurodegenerative disease research, the company’s drug candidate, GT-02287, has exhibited promising results in preclinical studies and clinical trials. The potential impact of GT-02287 extends beyond Parkinson’s disease (PD) to include other neurodegenerative disorders. The underlying science behind GT-02287’s mechanism of action has garnered attention for its potential to address a wide range of debilitating conditions, making it a focal point of interest for investors and stakeholders alike.
First-patient Dosage Achievement in GBA1 Parkinson’s Patients
In a significant pacesetting move, Gain Therapeutics recently announced the commencement of dosing for the first two participants in a Phase 1 clinical trial for GT-02287 targeting GBA-1 PD. This randomized, double-blind trial is designed to assess the drug’s safety, tolerability, as well as its impact on lysosomal GCase. The company’s intention to add a cohort of Parkinson’s patients to the trial post-MAD phase completion signifies a crucial step towards establishing clinical proof of concept based on biomarkers of the GCase cascade, which could significantly enhance the program’s value and standing within the biopharmaceutical landscape.
Expansion Potential for GT-02287
Gain’s strategic approach to focus on patient subgroups with biological compatibility for its drug’s MoA reflects a discerning investment in precision medicine. By aligning their therapeutic pursuits with targeted patient populations, the company aims to mitigate the risks typically associated with broad-spectrum treatments. Furthermore, the potential implications of GT-02287 beyond GBA1-PD exemplify the drug’s versatility and could significantly augment its market outlook. The identification of neuroprotective and motor function recovery effects in a Parkinson’s model hints at the broader therapeutic potential that GT-02287 holds, exemplifying its potential as a transformative agent in the treatment landscape.
GT-02287 and the Prospect of Broadening Therapeutic Indications
As scientific evidence suggests the potential applicability of GT-02287 across a spectrum of neurodegenerative disorders, the drug’s capacity to enhance lysosomal GCase enzyme activity not only in PD but also in other related conditions like Gaucher disease underscores its significance as a versatile therapeutic candidate. With the potential to expand its addressable market from $1 billion to $5 billion in peak sales, GT-02287’s relevance in addressing the unmet medical needs of millions affected by neurodegenerative diseases becomes increasingly apparent.
GT-02287: A Glimmer of Hope for Gaucher Disease
In its pursuit of addressing lysosomal storage disorders, Gain’s exploration of the therapeutic potential of GT-02287 in Gaucher disease offers a ray of hope for individuals grappling with this prevalent condition. By leveraging the drug’s ability to restore lysosomal function and mitigate the buildup of glucosylceramide, Gain Therapeutics seeks to provide a compelling treatment option for individuals affected by this challenging disorder.
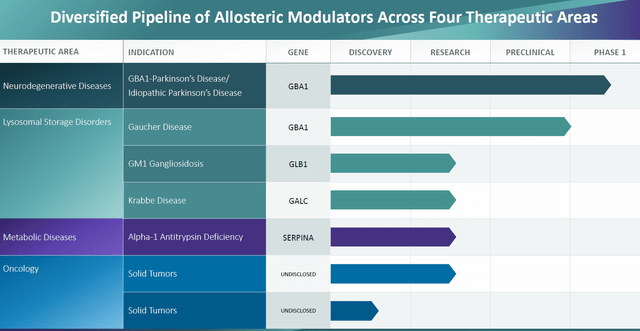
Exploring Gain’s Diversified Pipeline
Gain’s commitment to advancing therapeutic solutions extends beyond neurodegenerative disorders, with the company’s efforts directed towards addressing an array of lysosomal storage disorders. The promising preclinical findings surrounding compounds targeting GM1 gangliodosis and alpha-1 antitrypsin deficiency reflect Gain’s multifaceted approach towards developing transformative treatments across a spectrum of debilitating conditions, underscoring their dedication to pioneering innovative solutions in the biopharmaceutical landscape.
Assessing Gain Therapeutics’ Potential in the Current Biopharmaceutical Landscape
Revolutionizing Lysosomal Treatment: The Influence of Biogen’s Alectos partnership and Merck’s Caraway acquisition
Biogen’s noteworthy partnership with Alectos for the selective GBA2 inhibitor and Merck’s striking acquisition of Caraway Therapeutics signaled the industry’s deepening focus on the lysosome’s critical role in combating neurodegenerative diseases, including Parkinson’s Disease (PD). The cascade of scientific advancements align with Gain’s strategic position in this burgeoning therapeutic domain.
Big Pharma Frenzy: AI-driven Companies Garnering Unprecedented Interest
Big pharma’s burgeoning investment in AI-driven companies, skyrocketing to $60 billion in March 2023, heralds a significant shift towards artificial intelligence as a driving force in pharmaceutical innovation. This mounting interest underscores the potential for Gain’s proprietary drug discovery engine, paving the way for prospective endeavors with prominent stakeholders.
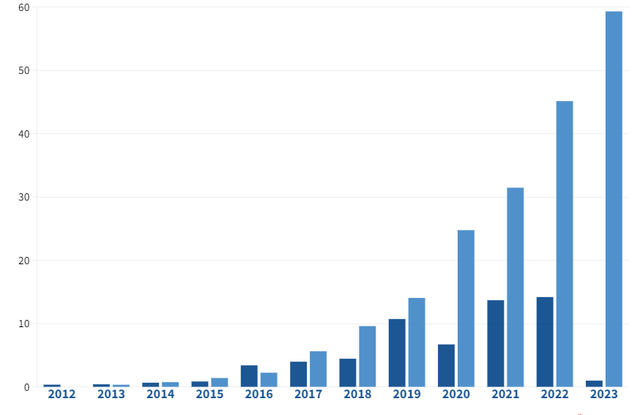
The anticipated integration of AI in drug development, with an estimated 50% involvement by 2030, accentuates AI’s pivotal role as a priority for major pharmaceutical companies, setting the stage for promising prospects for entities like Gain.
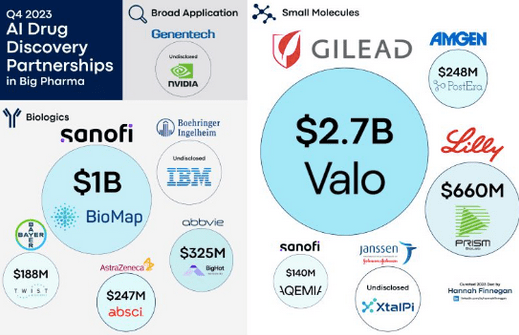
Amid the fervent momentum surrounding AI, Gain’s potential to leverage this wave of interest within the pharmaceutical sphere is genuinely promising.
Evaluating Financial Footing: A Deeper Dive into Gain’s Fiscal Position
As of September 30, 2023, Gain reported a cash balance of $12.3 million, with the subsequent closure of a $10.1 million public offering and private placement further bolstering its financial resilience. This financial backing, featuring notable investors like Andrew Schwartzberg and David Einhorn’s Greenlight Capital, underscores the market’s confidence in Gain’s distinctive trajectory.
Such substantial financial backing, coupled with sophisticated investors, reinforces Gain’s solidity in navigating its ongoing clinical trials and pioneering initiatives within the biopharmaceutical landscape.
Navigating Risks: Cautionary Assessment of Potential Hurdles
Gain’s early-stage status and the inherent regulatory uncertainties pose potential challenges, accentuating the need for cautious optimism. Moreover, unanticipated scientific developments or competitive dynamics could significantly impact Gain’s trajectory, necessitating vigilance amid the company’s promising prospects.
Undervalued Potential: Rethinking Gain’s Market Position
With a market cap of $55 million, Gain’s valuation appears remarkably modest in light of its pioneering assets like GT-02287 and its AI-driven drug discovery platform. The converging industry dynamics underscore the intrinsic potential of Gain’s assets and reinforce its undervalued market position, setting the stage for prospective growth and acclaim.
In Closing: Charting Gain’s Trajectory Amid Unprecedented Potential
Gain’s strategic positioning, underscored by its groundbreaking asset GT-02287 and distinctive AI-driven drug discovery platform, unfurl a realm of unparalleled potential within the biopharmaceutical landscape. The compelling convergence of scientific breakthroughs and financial backing cements Gain’s noteworthy standing, signaling a promising trajectory amidst a fervent industry landscape.
Gain’s unique position within the biopharmaceutical domain, accentuated by its promising assets and strategic alliances, underscores an enticing narrative for prospective stakeholders. The company’s steadfast endeavors in combating neurodegenerative diseases and innovating within the AI-driven drug discovery sphere set a dynamic stage for future growth and acclaim, carrying substantial potential amidst a profoundly transformative industry landscape.
Editor’s Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.