Revolutionizing Treatment for Rare Diseases
Intellia Therapeutics, Inc., known by its ticker symbol NTLA, has initiated dosing for the first patient in the ambitious phase III MAGNITUDE study. The study aims to evaluate the safety and efficacy of NTLA-2001, the company’s investigational in vivo genome-editing candidate designed to combat transthyretin (ATTR) amyloidosis.
An Eager Dive into the Unknown
Intellia’s leap into the phase III MAGNITUDE study comes on the heels of promising interim results from a phase I trial. These initial findings hinted at substantial reductions in the TTR protein, a prime culprit in the development of ATTR amyloidosis, following treatment with a single dose of NTLA-2001.
From Setback to Comeback:
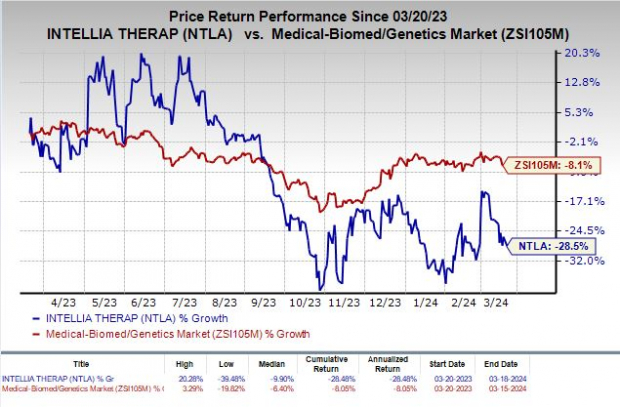
Despite a challenging year, with Intellia’s shares dropping by 28.5%, the company’s recent progress in the MAGNITUDE study has reignited hope among investors. Success in this pivotal study could potentially pave the way for regulatory approvals on a global scale.
The First of Its Kind:
Should NTLA-2001 receive approval, it would mark a historic milestone as the maiden gene editing treatment for ATTR amyloidosis. Intellia’s journey with NTLA-2001, the company’s debutant clinical candidate, represents a significant stride in the biopharmaceutical realm.
Collaborative Ventures and Future Prospects
In this endeavor, Intellia has joined forces with Regeneron Pharmaceuticals (REGN), forging an alliance aimed at fostering the development and commercialization of NTLA-2001. While taking the lead on NTLA-2001, Intellia and Regeneron are set to jointly share costs and profits in a partnership that underscores the industry’s collaborative spirit.
Fanning the Flames of Possibility:
Beyond its present focus on ATTR amyloidosis with cardiomyopathy, Intellia also harbors plans to assess NTLA-2001’s effectiveness in treating hereditary ATTR amyloidosis with polyneuropathy. Initiating a pivotal phase III study in this supplementary indication signals the company’s unwavering commitment to combating a spectrum of rare diseases.
Shifting the Paradigm: A Glimpse Into a Brighter Tomorrow
As the healthcare landscape evolves, Intellia’s strides in genome editing exemplify the industry’s tireless pursuit of innovative solutions. With NTLA-2001 steering the company towards uncharted territories, investors and patients alike watch with bated breath as the narrative unfolds.