A Prescription For Success:
Johnson & Johnson has hit a milestone as an FDA committee overwhelmingly supports broadening the scope of Carvykti, its innovative multiple myeloma drug. The committee’s nod comes as J&J seeks to widen Carvykti’s application to include adult patients facing relapsed or refractory multiple myeloma.
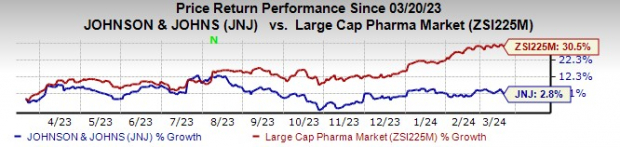
After scrutinizing data from the phase III CARTITUDE-4 study – the backbone of the supplemental biologics license application (sBLA) – the FDA’s Oncologic Drugs Advisory Committee slam-dunked its favorable opinion with an 11-0 vote. This commendation follows on the heels of a turbulent year that saw J&J’s stocks eke out a modest 2.8% gain in comparison to a heftier 30.5% surge seen in the pharmaceutical industry.
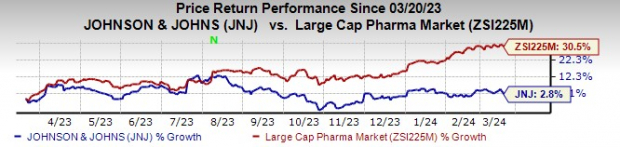
Image Source: Zacks Investment Research
The CARTITUDE-4 trial vividly demonstrated the drug’s prowess by achieving a statistically significant improvement in progression-free survival. Patients on Carvykti stood a 74% lower risk of the disease advancing compared to current standard treatments. The much-anticipated FDA verdict on the sBLA is slated for April 5.
Carvykti presently caters to adults tackling relapsed or refractory multiple myeloma after exhausting four or more prior lines of therapy, involving a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
J&J also has its sights set across the pond, having filed a Type II variation application with the European Medicines Agency. This application seeks to expand Carvykti’s label for treating adult patients grappling with relapse and lenalidomide-refractory multiple myeloma. Last month, the Committee for Medicinal Products for Human Use gave the green light by offering a favorable opinion recommending approval for Carvykti’s extended usage.
Investment Insights:
J&J currently stands at a Zacks Rank #3 (Hold).
Charting Success:
Check out the latest insights on the performance of Johnson & Johnson here.
When it comes to healthcare investments, keep an eye on Zacks Rank #1 (Strong Buy) companies like Vanda Pharmaceuticals, ADMA Biologics, and GSK, plc.
Having shown promising growth in the past 60 days, these companies are ones to watch for potential gains in the healthcare sector. Each boasting significant improvements in earnings estimates, they offer investors a favorable outlook for the future.
While Vanda Pharmaceuticals’ shares experienced a downturn of 42.3% over the past year, the company delivered an impressive three-quarter average earnings surprise of 92.88%. ADMA Biologics, on the other hand, watched its shares soar by 94.5%, with an outstanding four-quarter average earnings surprise of 85.0%. Meanwhile, GSK saw a respectable 20.8% uptick in shares, with a solid four-quarter average earnings surprise of 7.59%.
Interesting in spotting growth opportunities in the infrastructure sector? A major revamp is on the horizon, promising significant returns for savvy investors.
Get ahead of the curve by delving into Zacks’ Special Report, offering insights into five companies poised to benefit from the upcoming construction and repair boom. The potential for massive growth is tangible, so why wait? Grab your free copy today.
Download FREE: How To Profit From Trillions On Spending For Infrastructure >>
Access the latest recommendations from Zacks Investment Research with a free report here.
Uncover insights into GSK PLC Sponsored ADR (GSK) with a Free Stock Analysis Report.
Eager to know more about Johnson & Johnson (JNJ)? Examine our Free Stock Analysis Report here.
Discover the insights into Vanda Pharmaceuticals Inc. (VNDA) with a Free Stock Analysis Report.
Gain valuable information about ADMA Biologics Inc (ADMA) with a Free Stock Analysis Report.
For the full article on this exciting development, click here.
Dive deeper into Zacks Investment Research
Keep on top of Nasdaq, Inc.’s viewpoints through careful examination and reflection.