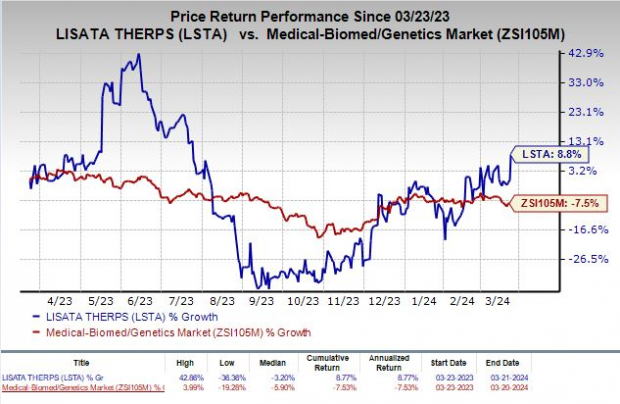
Steering into Uncharted Territory: RPDD Tag for Osteosarcoma
In a landmark development, Lisata Therapeutics, Inc. has made waves by securing the Rare Pediatric Disease Designation (“RPDD”) from the FDA for its groundbreaking pipeline asset, LSTA1. This cutting-edge therapy has set its sights on revolutionizing the treatment landscape for osteosarcoma, a rare bone cancer predominantly striking children, adolescents, and young adults. The bullish response was evident, with a notable 8.4% surge in the company’s stock on Mar 21, in the wake of this seminal announcement.
Charting New Frontiers: The Impact of RPDD
The RPDD badge of honor, bestowed upon therapies addressing severe or life-threatening illnesses impacting less than 200,000 minnows in the United States, marks a strategic victory for Lisata. With an eventual FDA seal of approval, the company could potentially snag a priority review voucher (“PRV”), unlocking avenues for future growth. The PRV, akin to a golden ticket, could be utilized by Lisata for subsequent FDA submissions or even traded to bolster financial reserves, adding a lucrative twist to its repertoire.
Riding the Crest: LSTA1’s Therapeutic Voyage
LSTA1, the trailblazing investigational compound, brings a novel paradigm to the table by enhancing the infiltration of partnered anti-cancer medications into solid tumor strongholds. By activating an innovative uptake pathway, this candidate has shown promise in transforming the tumor microenvironment, priming tumors for increased receptivity towards immunotherapy treatments.
The metamorphosis of Lisata on the stock exchange is equally momentous, with a commendable 8.8% uptick in the past year, defying the industry downtrend of 7.5%.
Expansive Frontiers: LSTA1’s Multi-Faceted Application
Embracing a multi-pronged approach, LSTA1 is under the microscope in diverse anti-cancer regimens across various cancer types in early-to-mid-stage clinical investigations. The canvas includes metastatic pancreatic ductal adenocarcinoma (“mPDAC”), diverse pancreatic cancers, cholangiocarcinoma, esophageal cancer, head and neck cancer, appendiceal cancer, colon cancer, and glioblastoma multiforme (“GBM”).
The nod of approval from the FDA with an orphan drug designation for treating pancreatic cancer and GBM lends further credence to the far-reaching impact of LSTA1 on cancer therapeutics.
Trailblazing the Future: Milestones and Beyond
Setting the stage for future triumphs, Lisata achieved a significant milestone by initiating a phase IIa study in January 2024, investigating LSTA1 in combination with the gold standard, temozolomide, for newly diagnosed GBM patients. Moreover, the completion of patient enrollment in the phase IIb ASCEND trial in December 2023, focusing on LSTA1’s collaboration with gemcitabine/nab-paclitaxel in first-line mPDAC patients, underscores the company’s unwavering commitment to medical breakthroughs.
With top-line data from ASCEND cohort A on the horizon, anticipated by the year’s end, and full dataset disclosure slated for mid-2025, Lisata shows no signs of slowing down its quest to carve a niche in the fiercely competitive oncology landscape.