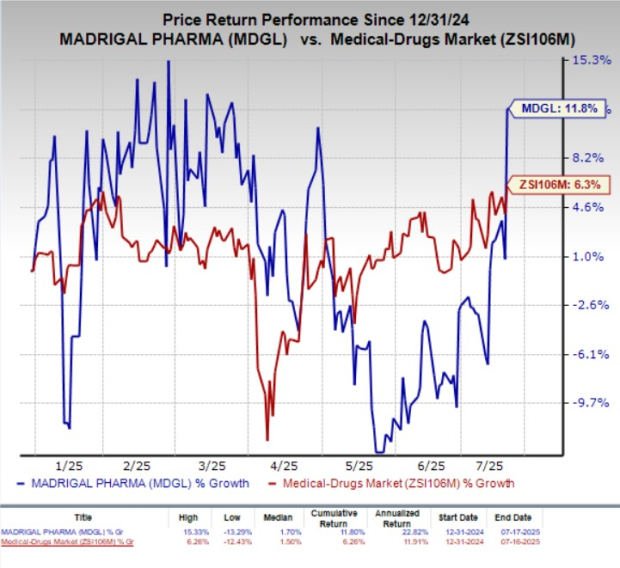
Madrigal Pharmaceuticals (MDGL) shares increased by 10.9% on Wednesday following a Notice of Allowance for a new U.S. patent related to its FDA-approved weight-based dosing regimen for Rezdiffra (resmetirom). This patent, set to protect the drug until September 30, 2044, aims to enhance market exclusivity by preventing generic versions from entering the market.
Rezdiffra, which received accelerated FDA approval in 2024, is the first approved therapy for metabolic dysfunction-associated steatohepatitis (MASH) in adults with moderate to advanced liver fibrosis. Currently, over 17,000 patients are being treated with Rezdiffra, demonstrating strong demand since its commercial launch in the U.S.
Madrigal’s regulatory filing for the MASH indication is under review in the EU, with a decision expected in August 2025. Ongoing studies, including the MAESTRO-NASH and MAESTRO-NASH OUTCOMES trials, aim to provide additional data supporting Rezdiffra’s full approval.