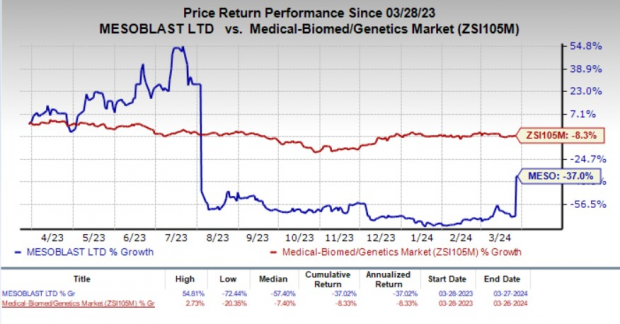
Mesoblast Limited, known by ticker symbol MESO, witnessed a staggering 78.6% surge on March 26. This uptick came on the heels of a promising development from the FDA regarding their primary product candidate, remestemcel-L. This drug is on a trajectory to target a dire medical need – the treatment of pediatric patients grappling with steroid-refractory acute graft versus host disease (SR-aGVHD).
Following a thorough review, the FDA informed Mesoblast that the clinical data from the phase III MSB-GVHD001 study on remestemcel-L meets the threshold for resubmission of the biologics license application (BLA) catered to pediatric patients with SR-aGVHD.
Mesoblast is now actively preparing to submit the BLA once again, tentatively scheduled for the second quarter of 2024, with a focused approach to address the remaining product characterization issues. Interestingly, in the absence of any approved therapies for youngsters under 12 afflicted with aGVHD in the U.S., MESO stands on the brink of a significant market opportunity, awaiting the FDA’s nod on the regulatory application for remestemcel-L in this particular area.
Over the past year, MESO shares took a hit, plunging by 37% while the broader medical biomed sector experienced an 8.5% dip.
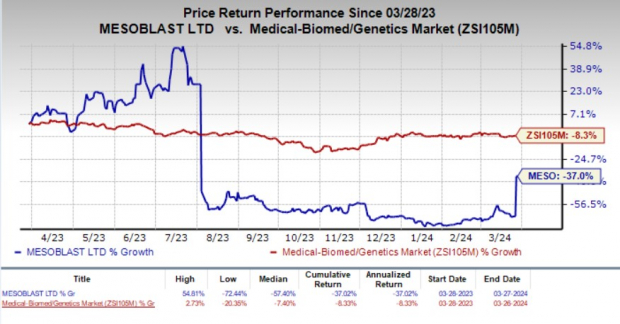
Image Source: Zacks Investment Research
Worth noting is that Mesoblast had faced FDA denial twice previously for marketing clearance of remestemcel-L for pediatric SR-aGvHD. The regulatory authority cited shortcomings in clinical and manufacturing data, urging additional data to back the approval of remestemcel-L in this context, including potency assay or other clinical data to fortify the case.
Additionally, Mesoblast has teamed up with the Blood and Marrow Transplant Clinical Trials Network (BMT CTN) to embark on a pivotal phase III trial of remestemcel-L targeting adults with SR-aGVHD.
This collaboration with the BMT CTN, a network comprising centers that collectively conduct nearly 80% of U.S. allogeneic blood and marrow transplants, is backed by the United States National Institutes of Health.
The upcoming pivotal late-phase study on remestemcel-L will scrutinize the drug’s effectiveness in patients aged 12 years and above who have proven unresponsive to corticosteroids and a second-line agent like Incyte Corporation’s INCY Jakafi (ruxolitinib). Presently, there are no approved treatments for this specific patient cohort.
In parallel, MESO is actively involved in advancing remestemcel-L in mid to late-stage trials focusing on two more indications – inflammatory bowel disease and acute respiratory distress syndrome. Furthermore, another product candidate in the pipeline, rexlemestrocel-L, is being groomed for the treatment of advanced chronic heart failure and chronic low back pain.
Incyte’s Jakafi, a pioneering JAK1/JAK2 inhibitor, remains the solitary FDA-approved remedy for SR-aGvHD across both adult and pediatric patients aged 12 years or older in the U.S. Notably, INCY has partnered with Swiss pharmaceutical titan Novartis NVS for the global distribution of Jakafi. It’s essential to underline that while Incyte markets Jakafi in the U.S., Novartis handles its global presence under the name Jakavi.
Jakafi earned its initial FDA approval for treating patients with polycythemia vera (PV) who demonstrated inadequate response or intolerance to hydroxyurea. It’s also sanctioned for patients exhibiting intermediate or high-risk myelofibrosis (MF), including primary MF, post-PV MF, and post-essential thrombocythemia MF.
Charting Mesoblast Limited Price and Consensus
Mesoblast Limited price-consensus-chart | Mesoblast Limited Quote
Expert Insight and Top Pick
As of now, Mesoblast boasts a Zacks Rank #4 (Sell).
A notable alternate stock of interest is ADMA Biologics ADMA, which holds an impressive Zacks Rank #1 (Strong Buy) currently. You can dive into today’s full roster of Zacks #1 Rank stocks here.
Over the last 30 days, the Zacks Consensus Estimate for ADMA Biologics’ 2024 earnings per share (EPS) has surged from 22 cents to 30 cents. Meanwhile, the forecast for ADMA’s 2025 EPS has skyrocketed from 32 cents to 50 cents during the same timeframe. ADMA shares have notably climbed by 102.5% in the past year.
ADMA pulled off earnings surprises in three of the past four quarters with an average earnings beat of 85%.
Discover 7 Top Stocks for the Upcoming Month
The latest list reveals 7 premier stocks handpicked from a pool of 220 Zacks Rank #1 Strong Buys. These stocks are dubbed as “Most Likely for Early Price Pops.”
Since 1988, this exclusive list has outperformed the market with an impressive annual gain of over +24.2%. So, set your sights on these elite 7 for potential swift growth.
The views and opinions shared here are solely those of the author and may not align with Nasdaq, Inc.