Moderna’s mNexspike COVID-19 Vaccine Approved by FDA
Moderna (MRNA) has secured FDA approval for mRNA-1283, its next-generation refrigerator-stable COVID-19 vaccine, now branded as mNexspike. The approval, however, is more limited than anticipated.
Approval Details and Target Population
The FDA’s recent guidance focuses on vaccinating high-risk groups. mNexspike is approved for individuals aged 65 and older, as well as those aged 12-64 with at least one underlying health condition. Initially, Moderna sought broader approval for all individuals aged 12 and older.
Product Portfolio Expansion
With this approval, mNexspike becomes Moderna’s third FDA-approved product, joining Spikevax and mResvia. Spikevax is authorized for use in those aged 6 months and older.
Vaccine Efficacy and Launch Plans
Moderna claims that mNexspike offers an improved shelf life and storage advantages, which are crucial for distribution in areas with limited cold-chain infrastructure. Data from a late-stage study indicated that mNexspike is at least as effective as Spikevax, showing a 9.3% higher relative efficacy with a 10 μg dose compared to a 50 μg dose of Spikevax. Moderna plans to launch mNexspike commercially in the 2025-26 vaccination season.
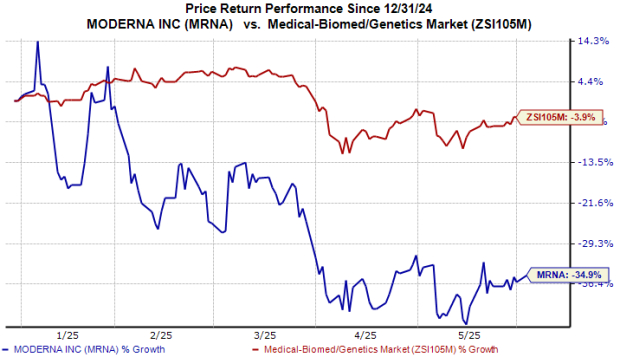
MRNA Stock Performance
This year, MRNA shares have declined by 35%, compared to a 4% drop in the broader industry.
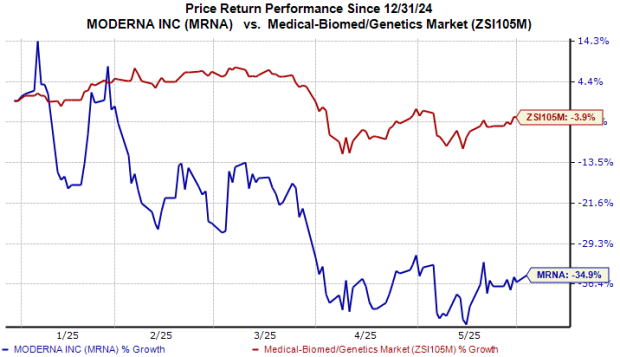
Image Source: Zacks Investment Research
Current Challenges for Moderna’s Stock
Recent developments have negatively impacted MRNA’s stock sentiment. The U.S. government terminated a $766 million contract with Moderna for its bird flu vaccine’s late-stage development, affecting its finances as the company seeks to cut operating costs.
Additionally, new FDA COVID-19 booster guidelines limit eligibility for healthy individuals under 65, which may reduce demand for vaccines. Moreover, Moderna voluntarily withdrew its FDA filing for a COVID-19/influenza combination vaccine, seeking more efficacy data for the flu component, delaying potential approval.
Competitive Landscape in COVID-19 Vaccines
Other key players include Pfizer (PFE) and Novavax (NVAX), both of which have their own COVID-19 vaccines: Comirnaty and Nuvaxovid, respectively. Pfizer’s vaccine, based on mRNA technology developed with BioNTech, shares similar approval criteria with Spikevax.
In contrast, Novavax’s Nuvaxovid, the only non-mRNA vaccine approved for older adults and high-risk individuals, recently received full FDA approval but is not yet available for those under 12.
MRNA’s Stock Rank
Currently, Moderna holds a Zacks Rank #3 (Hold).
Disclaimer
The views and opinions expressed herein are those of the author and do not necessarily reflect those of Nasdaq, Inc.