Novo Nordisk (NVO) announced the FDA’s approval of Wegovy HD, a once-weekly injectable semaglutide 7.2 mg, aimed at reducing excess body weight and maintaining weight reduction long-term. The product is set for a U.S. launch in April 2026. This approval is notable as it is the first under the FDA’s Commissioner’s National Priority Voucher pilot program, designed to expedite reviews of treatments addressing urgent public health needs.
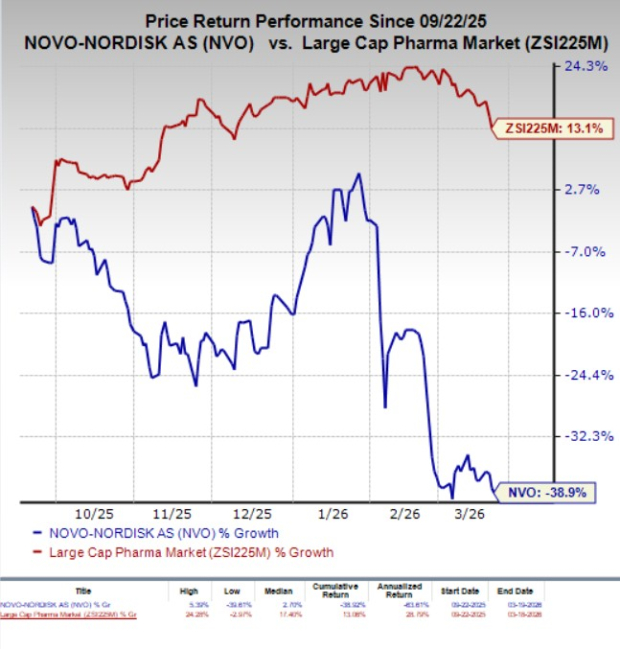
The Wegovy drug line, which includes various formulations, has demonstrated significant weight loss effectiveness, particularly the 7.2 mg dose, which achieved an average weight reduction of 20.7% over 72 weeks in clinical trials. Studies also revealed that approximately one in three patients on this higher dose attained at least a 25% weight loss. In the past six months, shares of Novo Nordisk have decreased by 38.9%, contrasting with a 13.1% growth in the industry.