QuidelOrtho Corporation recently celebrated the FDA clearance for its QuickVue COVID-19 test. This green light from the FDA, coupled with CLIA certificates of waiver, empowers the public to harness the test’s capabilities for home and medical healthcare settings.
The company shared a promising update on the Savanna platform, which obtained approval back in December 2023.
Stock Market Struggles
Despite these significant milestones, QuidelOrtho hasn’t been having the best run in the stock market. Over the last half-year, its shares plummeted by 43.1%, contrasting sharply with the industry’s rise of 24.3%. The broader S&P 500 index fared better with a gain of 19.5% during the same period.
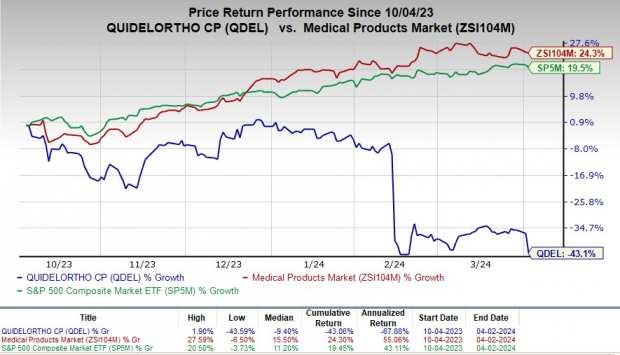
Image Source: Zacks Investment Research
Examining the QuickVue COVID-19 Test
Designed for symptomatic individuals within six days of showing symptoms, the QuickVue COVID-19 test brings a ray of hope. Its approval for self-administration in individuals aged 14 years and older, and administration by an adult for those aged two years and above, makes it a versatile tool in battling the pandemic. The test’s user-friendly instructions ensure easy integration into existing diagnostic processes.
Progress with the Savanna Platform
A crucial feather in QuidelOrtho’s cap is the green signal for the Savanna HSV 1+2/VZV PCR assay received in December 2023. This landmark approval empowers U.S. laboratories with moderate to high complexity in diagnostic testing to market and distribute the Savanna multiplex molecular platform alongside the Savanna HSV 1+2/VZV assay.
The company is actively working on the Savanna RVP4+ assay, which failed to meet expectations after a nine-month study on four viruses, leading to the withdrawal of its FDA regulatory submission in February. The focus has now shifted to developing the next-generation RVP4+ assay slated for commercial availability during the 2024/2025 respiratory season.
Additionally, plans are in motion for a sexually transmitted infection (STI) panel, with a clinical study expected to kick off in the second quarter.
Noteworthy Achievements
QuidelOrtho Corporation recently marked a significant milestone with Health Canada’s approval for its Triage PLGF test in Canada for laboratory use. This test aims to identify angiogenic imbalance, a condition linked to pregnancy complications like pre-eclampsia resulting from abnormal placentation and impaired placental blood flow.
Stock Analysis and Recommendations
With a Zacks Rank #5 (Strong Sell), QuidelOrtho faces certain hurdles in the current market scenario. However, amidst the ongoings, there’s good news in the medical industry as well. Companies like DaVita Inc., Cardinal Health, Inc., and Cencora, Inc., are shining examples with Zacks Rank #1 (Strong Buy) and Rank #2 (Buy) statuses, offering a beacon of hope to investors.
In this ever-evolving landscape, thorough stock analysis and industry trends can provide valuable insights for investors looking to navigate the complex and often unpredictable terrains of the medical sector.
Zacks Names #1 Semiconductor Stock
It’s only 1/9,000th the size of NVIDIA which skyrocketed more than +800% since we recommended it. NVIDIA is still strong, but our new top chip stock has much more room to boom.
With strong earnings growth and an expanding customer base, it’s positioned to feed the rampant demand for Artificial Intelligence, Machine Learning, and Internet of Things. Global semiconductor manufacturing is projected to explode from $452 billion in 2021 to $803 billion by 2028.
See This Stock Now for Free >>
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.