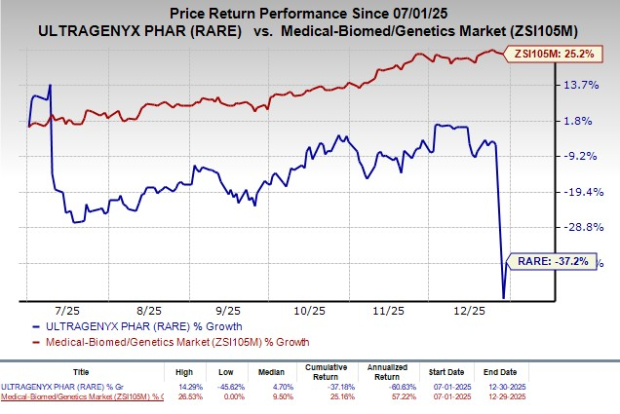
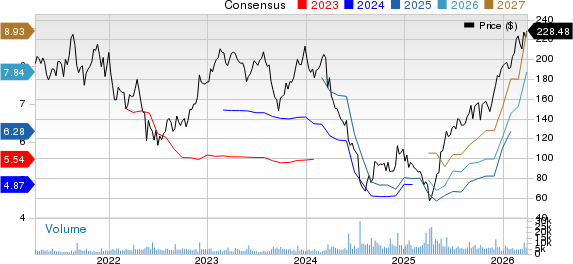
Ultragenyx Pharmaceutical (RARE) saw a 15.5% stock increase following the completion of a rolling submission of a biologics license application (BLA) to the FDA for its gene therapy DTX401 (pariglasgene brecaparvovec). This submission aims for approval to treat glycogen storage disease type Ia (GSDIa), a rare metabolic disorder impacting approximately 6,000 individuals globally.
The BLA is supported by data from a comprehensive clinical program, including the phase III GlucoGene study, which demonstrated a statistically significant 41% reduction in daily cornstarch intake after 48 weeks of treatment with DTX401 compared to a 10% reduction in the placebo group. Notably, if approved, DTX401 would be the first therapy addressing the underlying cause of GSDIa.
The rolling submission included chemistry, manufacturing, and controls modules, completed in August 2025. The company reported positive long-term data indicating improved blood glucose control and better fasting tolerance for patients at 96 weeks into the treatment.