Regeneron’s Unfortunate Regulatory Hurdle
Regeneron Pharmaceuticals, Inc. (REGN) encountered a stumbling block as the FDA delivered Complete Response Letters (CRLs) for its biologics license application (BLA) regarding odronextamab – an investigational bispecific monoclonal antibody targeted at relapsed/refractory follicular lymphoma (FL) and relapsed/refractory diffuse large B-cell lymphoma (DLBCL), specifically after multiple lines of systemic therapy.
Reason for Rejection
The setback was not due to any clinical efficacy or safety concerns but rather related to enrollment status in the confirmatory trials. This mishap has hit Regeneron’s strategic aspirations of broadening its oncology arsenal, which presently boasts the antibody Libtayo.
The Impact and Ongoing Efforts
The rejection delays potential approval and dampens REGN’s efforts to revolutionize B-cell non-Hodgkin lymphoma treatment under the OLYMPIA program. While patient enrollment has commenced for dose-finding studies, the confirmatory phase needs quick initiation to meet FDA’s approval criteria.
Future Prospects and Regulatory Timeline
Regeneron aims to provide updates on enrollment progress and regulatory timelines towards the end of 2024. Concurrently, odronextamab awaits review in the European Union for R/R DLBCL and R/R FL with Orphan Drug Designation in the EU for these indications.
Stock Performance and Diversification Efforts
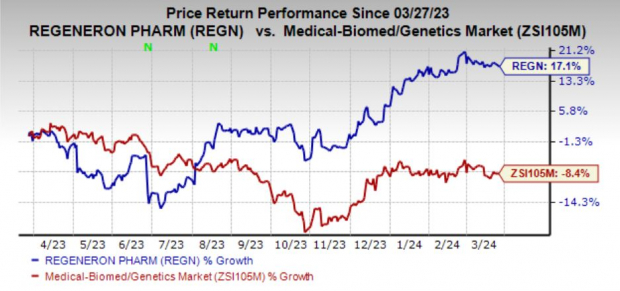
Despite the setback, Regeneron has witnessed a 17.1% surge in shares over the past year. The company is also driving growth through the expanding usage of Libtayo across multiple cancer types and acceptance of linvoseltamab for Priority Review by the FDA to treat relapsed/refractory multiple myeloma.
Conclusion and Industry Outlook
This regulatory hiccup is a reminder of the unpredictable nature of drug development and underscores the challenges faced by pharmaceutical companies in securing regulatory approvals.
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.