The reality of over 1.1 million overdose deaths since 2000 looms large, a chilling trend that underscores the urgent need for safer pain management solutions. The specter of opioid addiction casts a long shadow on post-surgical recovery, with thousands ensnared in dependency each year.
Enter the prospect of a non-addictive pain remedy – a beacon of hope akin to the meteoric rise of GLP-1 weight-loss drugs. One company that might soon make this a reality is Vertex Pharmaceuticals.
Vertex Pharmaceuticals: Pioneering an Opioid Alternative
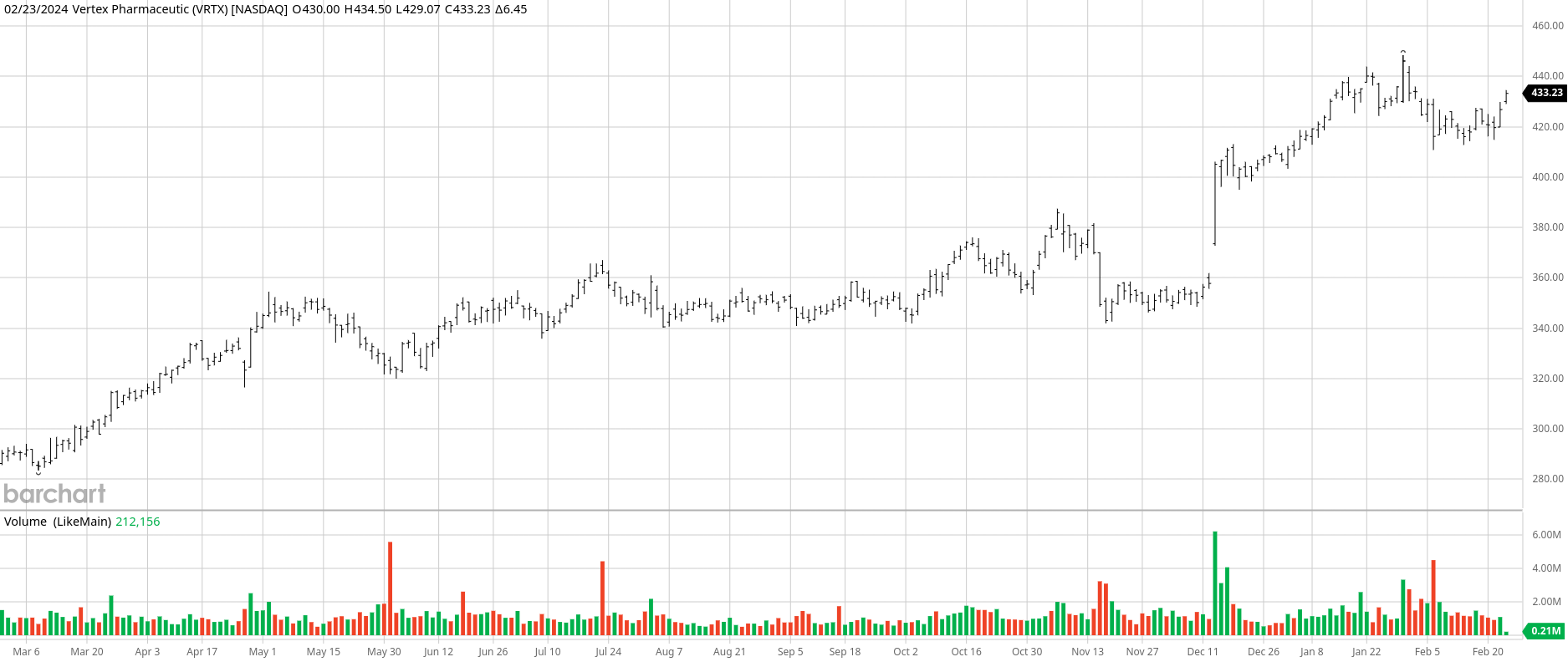
Vertex Pharmaceuticals (VRTX), a stalwart in the S&P 500 Index with a robust $110 billion market cap, made waves on Jan. 30 with the triumphant news of VX-548. This non-opioid pain reliever aced three Phase III trials, showcasing remarkable efficacy in alleviating pain across diverse scenarios.
While the drug demonstrated notable pain reduction post abdominoplasty and bunionectomy surgeries, it fell short of outperforming a common opioid blend in a critical secondary objective. This setback, however, does little to dim Vertex’s overall success.
The Nexus of Pain Relief: Inside VX-548
In a landscape fraught with opioid pitfalls, VX-548 shines as a rare beacon of promise. Vertex’s painstaking research on sodium channels culminated in the birth of this breakthrough drug, which targets Nav1.8 to stifle pain signals at the cellular level.
While not a panacea, VX-548 navigates the minefield of pain management, eschewing the shackles of opioid addiction that haunt traditional remedies. Vertex’s aspirations for VX-548 extend beyond its cystic fibrosis domain, marking a decisive push into broader therapeutic frontiers.
The Appeal of Vertex Stock
Vertex’s Trikafta/Kaftrio regimen continues to rake in substantial revenues, spotlighting its prowess in cystic fibrosis treatment. With plans to broaden CF patient coverage and expand product accessibility globally, Vertex remains a beacon of innovation and progress.
New horizons beckon for Vertex, with ventures into sickle cell disease and Type 1 diabetes expanding its therapeutic footprint. The recent FDA greenlight for Casgevy, a gene-editing marvel created in partnership with Crispr Therapeutics AG, underscores Vertex’s cutting-edge approach to medical innovation.
As Vertex steers toward securing approval for expanded indications and enhancing its drug portfolio, the monumental potential of VX-548 becomes increasingly evident. Vertex’s CEO, Reshma Kewalramani, champions the drug’s capacity to transform the acute pain landscape, heralding a seismic shift in therapeutic norms.
With VX-548 poised to capture a market teeming with acute pain sufferers, Vertex stands on the cusp of a groundbreaking chapter in pharmaceutical history. The future holds the promise of a blockbuster drug, setting Vertex on a trajectory towards sustained growth and success.

On the date of publication, Tony Daltorio had a position in: VRTX . All information and data in this article is solely for informational purposes. For more information please view the Barchart Disclosure Policy here.
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.