“`html
This week Novartis (NVS) improved its sales growth forecast for the upcoming years. The European Commission approved Pfizer’s (PFE) hemophilia medication, Hympavzi (marstacimab). Additionally, AstraZeneca (AZN) received a recommendation for the expanded use of Tagrisso in treating lung cancer in the EU. Both Johnson & Johnson (JNJ) and Lilly (LLY) reported favorable outcomes from their respective clinical studies.
Here’s a summary of the week’s key developments.
Novartis Boosts Sales Forecast
Novartis has raised its mid-term sales growth outlook, adjusting its compound annual growth rate (CAGR) target for the 2023-2028 timeframe from 5% to 6%. The company anticipates a sales CAGR of 5% during 2024-2029, starting from 2024 as the base year. This optimistic forecast relies on heightened expectations for significant drugs like Pluvicto, Kesimpta, Kisqali, Cosentyx, and Leqvio, alongside upcoming product launches. Novartis believes eight of its drugs may reach peak sales of between $3 billion and $8 billion. Furthermore, with 30 candidates in its pipeline, the company expects to maintain mid-single-digit sales growth beyond 2029. NVS remains focused on achieving a core operating income margin of over 40% by 2027.
Pfizer Secures EU Approval for Hemophilia Treatment
The European Commission has granted marketing authorization for Pfizer’s Hympavzi (marstacimab), aimed at treating hemophilia A and B. This therapy is designed to prevent or lessen the frequency of bleeding episodes in adults and adolescents with hemophilia who do not have inhibitors. The approval was supported by promising results from the phase III BASIS study. Hympavzi was already approved by the FDA in the United States last month.
This marks Pfizer’s second hemophilia drug approval in 2024, following the FDA’s green light for Beqvez/Durveqtix (fidanacogene elaparvovec), a one-time gene therapy for hemophilia B, which is still under review in Europe.
Positive Data for J&J’s Psoriasis Drug Icotrokinra
Johnson & Johnson announced that its pivotal phase III trial for icotrokinra (JNJ-2113) in treating moderate to severe plaque psoriasis successfully met its main goals at week 16. The ICONIC-LEAD study aimed to evaluate the Psoriasis Area and Severity Index (PASI) 90 and Investigator’s Global Assessment (IGA) of 0/1 response at week 16. Results showed that 64.7% of participants treated with icotrokinra achieved clear or nearly clear skin, and 49.6% met the PASI 90 criteria.
The positive results continued to grow through week 24, with 74% reporting clear or almost clear skin and 64.9% achieving PASI 90. Additionally, another phase III study, ICONIC-TOTAL, testing once-daily icotrokinra, also met its primary endpoint at week 16.
AstraZeneca’s Tagrisso Gets CHMP Nod for Expanded Usage
AstraZeneca revealed that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency has recommended the approval of Tagrisso (osimertinib) to treat unresectable EGFR-mutated non-small cell lung cancer (NSCLC) following platinum-based chemotherapy. Tagrisso has been recommended specifically for patients with tumors featuring exon 19 deletions or exon 21 (L858R) mutations, based on favorable data from the phase III LAURA study. The FDA had approved Tagrisso for similar applications in September.
Tagrisso is already authorized in various countries for first-line treatment of EGFR-mutated NSCLC, as well as for other specific cases, including in combination with chemotherapy.
Lilly’s Cholesterol Drug Shows Promise in Phase II Trials
Lilly’s oral cholesterol medication, muvalaplin, has demonstrated efficacy in reducing lipoprotein(a) or Lp(a) levels in a 12-week phase II study. The drug significantly lowered elevated Lp(a) levels when compared to a placebo, achieving the trial’s primary endpoint by showing a notable percentage change in Lp(a) from baseline to week 12. Remarkably, the highest dose (240 mg) led to an 85.8% reduction in Lp(a) levels.
Muvalaplin utilizes a unique mechanism that interrupts the interaction between apolipoprotein(a) and apolipoprotein(b), preventing Lp(a) formation. Apolipoprotein(a) plays a crucial role in increasing the risk of cardiovascular incidents.
In the past five trading sessions, the NYSE ARCA Pharmaceutical Index decreased by 1.1%.
Large Cap Pharmaceuticals Industry 5-Year Return
Large Cap Pharmaceuticals Industry 5YR % Return
Here’s how the eight major pharmaceutical stocks performed over the last five days.
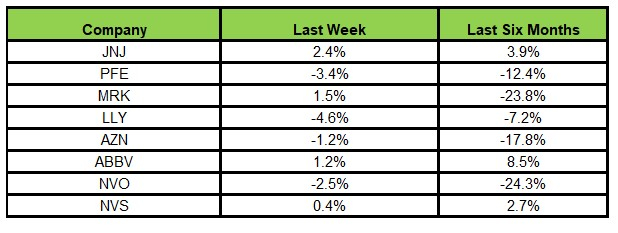
Image Source: Zacks Investment Research
During this period, Lilly experienced the largest decline at 4.6%, while J&J saw the biggest increase at 2.4%. Over the past six months, AbbVie reported the highest growth of 8.5%, while Novo Nordisk faced the steepest drop of 24.3%.
(For the latest updates on the pharmaceutical sector, see our previous recap: AZN, BAYRY’s Earnings, ABBV’s Pipeline Setback)
What’s Next for the Pharmaceutical Industry?
Stay tuned for regular updates on drug pipelines and regulatory changes in the coming week.
AstraZeneca, Lilly, Merck, and J&J hold a Zacks Rank #3 (Hold), while Pfizer is rated Zacks Rank #2 (Buy).
“`
Five Promising Stocks That Could See Massive Gains in 2024
Top Selections for Potential Growth
Experts at Zacks have identified five stocks as their top picks for the year ahead, each expected to gain +100% or more in 2024. While past recommendations have seen significant returns—such as +143.0%, +175.9%, +498.3%, and an impressive +673.0%—investors should remain cautious as not all picks guarantee success.
Many of the stocks mentioned are currently overlooked by major Wall Street investors, presenting a unique opportunity for those looking to invest early in potentially high-performing companies.
Discover Today’s Five Potential Home Runs >>
AstraZeneca PLC (AZN): Access our free stock analysis report.
Novartis AG (NVS): Get our free stock analysis report.
Johnson & Johnson (JNJ): View your free stock analysis report here.
Pfizer Inc. (PFE): Check out our free stock analysis report.
Eli Lilly and Company (LLY): Access your free stock analysis report.
For more details, read the full article on Zacks.com here.
The views and opinions expressed herein are those of the author and do not necessarily reflect those of Nasdaq, Inc.