Acadia Pharmaceuticals (ACAD) announced on October 4, 2023, that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has issued a negative opinion regarding the approval of trofinetide for treating Rett syndrome in patients aged two years and older in the EU. This decision follows an early February notice of a negative trend vote on Acadia’s marketing application, and the company plans to request a re-examination of the decision.
The pivotal LAVENDER study had met its primary and key secondary endpoints; however, the CHMP cited limitations in the treatment effect’s magnitude, insufficient symptom coverage in the study, and potential biases due to patient dropouts as reasons for the refusal. Rett syndrome affects approximately one in 10,000-15,000 female births globally, with symptoms typically emerging between six and 18 months of age.
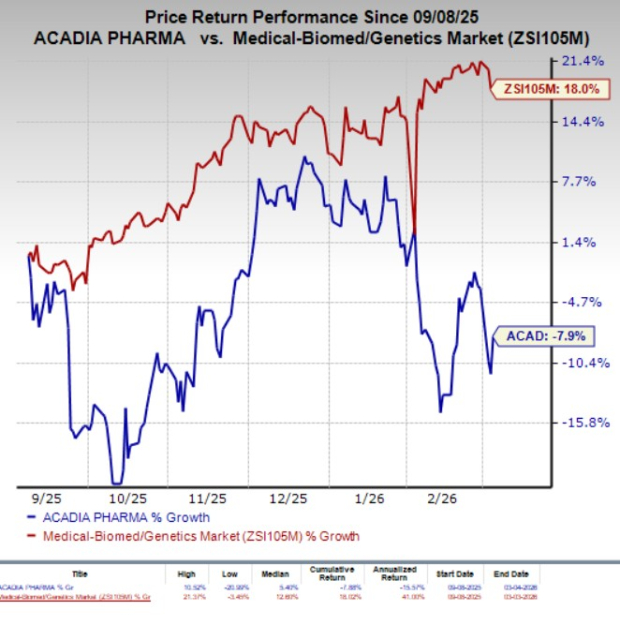
Despite the setback in Europe, Acadia’s lead products, Nuplazid and Daybue, are expected to generate around $1.7 billion in combined net sales by 2028. The FDA approved trofinetide as the first treatment for Rett syndrome in the U.S. in 2023, marketed as Daybue. In recent months, Acadia’s stock has decreased by 7.9%, compared to an 18% increase in the industry.