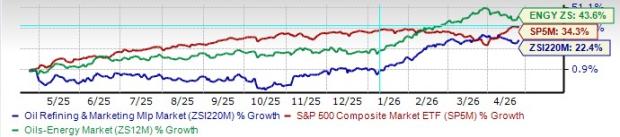
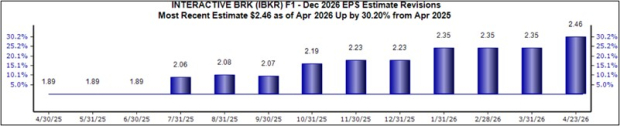
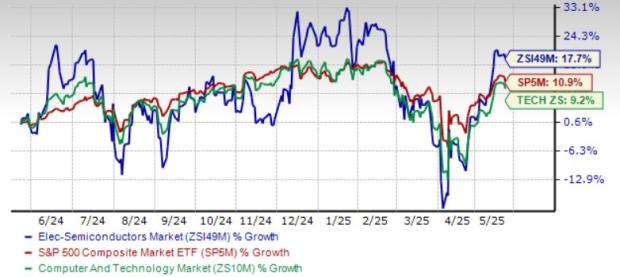
AstraZeneca and Daiichi Sankyo have received a monumental nod from the FDA with the acceptance of their biologics license application (BLA) for Dato-DXd for breast cancer treatment.
This groundbreaking application aims to address unresectable or metastatic HR+ HER2- breast cancer in patients who have undergone previous systemic therapy.
Future FDA Decision and Study Findings
By the first quarter of 2025, investors can anticipate the FDA’s decision on the application, given its standard review designation.
The BLA leans on the data from the TROPION-Breast01 phase III study, showcasing Dato-DXd’s significant advancement in progression-free survival compared to traditional chemotherapy.
Furthermore, the study noted promising trends in overall survival, though maturity is pending and will continue to be monitored.
If approved, Dato-DXd stands to offer patients an innovative alternative to conventional chemotherapy, heralding a new era in cancer treatment.
Global Impact and Industry Disruption
Beyond the FDA, AstraZeneca and Daiichi Sankyo are pursuing approvals for Dato-DXd in nonsquamous non-small cell lung cancer, expanding the potential of this revolutionary treatment.
This goes hand in hand with the global regulatory applications in the EU, China, and Japan, promising a far-reaching impact on cancer care worldwide.
Investor Perspective
In the healthcare sector, investors eyeing AstraZeneca might find reassurance in its Zacks Rank #3 (Hold) status.
Exploring Promising Healthcare Stocks
Among the standout healthcare stocks are Vanda Pharmaceuticals, ADMA Biologics, and MorphoSys, each boasting a Zacks Rank #1 (Strong Buy), offering tantalizing prospects to savvy investors.
Financial Performance
Detailed financial analysis reveals the stocks’ performance, with notable improvements and insights into earnings per share.
As the industry shifts towards ADCs like Dato-DXd for enhanced cancer treatment, AstraZeneca and Daiichi Sankyo’s pioneering efforts underscore a remarkable stride in battling cancer effectively.