Bristol Myers BMY shocked investors with news that the late-stage study on Zeposia (ozanimod) for Crohn’s disease (CD) did not meet its primary endpoint.
In the grand scheme of things, this was just the first of two induction studies in the phase III YELLOWSTONE clinical program, focusing on 600 adult patients with moderate to severe active CD over a 12-week treatment course. Regrettably, Zeposia fell short in achieving clinical remission at week 12, leaving stakeholders with a sour taste in their mouths.
Alas, no numerical data or figures were shared by Bristol Myers, leaving investors in the dark about the extent of the setback or the company’s next steps. Management has promised to unveil the full results in due course, likely at an upcoming medical meeting.
Beyond the induction studies, the YELLOWSTONE program encompasses a 52-week maintenance study and an optional 264-week, open-label extension (OLE) study.
CD, a chronic inflammatory bowel disease, inflicts sufferers with symptoms like diarrhea, abdominal pain, fatigue, and weight loss, making successful treatment paramount.
Year-to-date, Bristol Myers’ shares have edged up by 5.7%, outstripping the mere 0.5% uptick seen in the medical biomedicine and genetics industry.
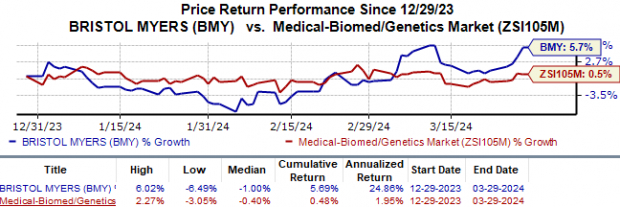
Image Source: Zacks Investment Research
Zeposia, a sphingosine 1-phosphate (S1P) receptor modulator, currently holds approval in the United States and Europe for treating multiple sclerosis (MS) and ulcerative colitis (UC).
However, the road ahead is fraught with challenges as Bristol Myers’ drug faces stiff competition from Pfizer’s S1P receptor modulator, Velsipity (etrasimod), which secured FDA approval last year for UC treatment. Not to mention, Pfizer is moving full steam ahead, evaluating etrasimod in multiple clinical stages for various immuno-inflammatory conditions.
Additionally, AbbVie’s JAK inhibitor Rinvoq received FDA nod for use in certain adult CD patients last year, further intensifying the competitive landscape for Bristol Myers.
While S1P receptor modulators like Zeposia and Pfizer’s Velsipity offer a fresh approach compared to existing UC therapies, both companies share the common pursuit of providing relief and remission to patients in need. Unfortunately, unlike Rinvoq and J&J’s Stelara, no S1P modulator has yet achieved late-stage success in treating CD.
In a separate development, Bristol Myers also disclosed positive results from the pivotal late-stage KRYSTAL-12 study, showcasing the efficacy of Krazati (adagrasib) in pre-treated patients with KRASG12C-mutated non-small cell lung cancer (NSCLC).
It’s worth noting that Krazati received accelerated approval from the FDA for treating adult patients with KRASG12C-mutated locally advanced or metastatic NSCLC. The KRYSTAL-12 study aims to cement this accelerated approval status with a full approval.
Bristol Myers Squibb Company Price
Bristol Myers Squibb Company price | Bristol Myers Squibb Company Quote
Zacks Rank & A Key Pick
Presently holding a Zacks Rank #3 (Hold), Bristol Myers pales in comparison to standout performers in the broader healthcare sector like ADMA Biologics. With a Zacks Rank #1 (Strong Buy), ADMA Biologics has been on a tear.
In the last 60 days, projections for ADMA Biologics’ 2024 earnings per share (EPS) have surged from 22 cents to 30 cents, followed by a robust increase in the estimates for 2025 from 32 cents to 50 cents. Meanwhile, ADMA’s stock has surged by a whopping 46.0% year-to-date.
Delivering earnings surprises in three out of the last four quarters, ADMA Biologics boasts an impressive four-quarter average earnings beat of 85.00%.
Just $1 to Discover All Zacks’ Picks
Believe it or not.
Back in the day, members were stunned when we offered them unrestricted access to our selections for a month, all for a single dollar. With no strings attached.
Thousands seized the chance, while many skeptics stayed on the sidelines, convinced there was a catch. And yes, there was a reason. We aimed to acquaint you with our suite of services like Surprise Trader, Stocks Under $10, Technology Innovators,and more. In 2023 alone, they successfully closed 162 positions with double- and triple-digit gains.
Bristol Myers Squibb Company (BMY) : Free Stock Analysis Report
Pfizer Inc. (PFE) : Free Stock Analysis Report
AbbVie Inc. (ABBV) : Free Stock Analysis Report
ADMA Biologics Inc (ADMA) : Free Stock Analysis Report
For more on this article, visit Zacks.com.
The sentiments outlined herein reflect the author’s personal views and not necessarily those of Nasdaq, Inc.