Roche has achieved a significant milestone with the FDA’s recent approval of the label expansion for Xolair (omalizumab), a drug initially intended to treat asthma. This approval opens new doors by extending treatment to help prevent allergic reactions, including anaphylaxis, in patients with IgE-mediated food allergies, marking a breakthrough for sufferers of this condition.
Positive Data from the OUtMATCH Study
The FDA’s approval is grounded in the positive findings from the OUtMATCH study. The research, which involved patients aged one to 55 with allergies to peanuts and at least two other food allergens, demonstrated that Xolair could significantly improve their ability to tolerate small amounts of triggering foods without an adverse reaction. This is a major boon for patients, showing that the treatment can enable young children as well as adults to consume these foods without fear of a severe allergic response, which is indeed a game-changer.
Statistical Insights
An indisputably outstanding finding was that a remarkably larger percentage of patients treated with Xolair could tolerate at least 600 mg of peanut protein without experiencing moderate to severe allergic symptoms compared to those who received a placebo. Moreover, the study showed that more patients treated with Xolair could tolerate significant amounts of protein from milk, egg, or cashew without encountering severe allergic symptoms in comparison to those on a placebo.
Impact and Market Considerations
This is a significant development, as it not only elevates the potential of Xolair but has the potential to positively impact Roche’s sales. The drug already holds approvals for the treatment of allergic asthma, chronic urticaria, and chronic rhinosinusitis with nasal polyps. With approximately 3.4 million children and 13.6 million adults in the US diagnosed with IgE-mediated food allergies, the approval of Xolair for food allergies addresses an unmet need in the market. This new approval is expected to be a key growth driver, especially when considering the lack of effective treatments currently available for food allergies.
Partnership and Competitive Landscape
Furthermore, Roche’s collaboration with Novartis on Xolair adds an interesting dynamic to the drug’s commercialization strategy. Roche co-promotes Xolair in the US, while Novartis takes charge of sales outside the country. This partnership is a strategic move that allows Roche to leverage both companies’ expertise to maximize market potential. It’s worth noting that the approval of Xolair for food allergies is occurring in the context of a competitive landscape, where Roche faces stiff competition from other players in the asthma space, such as Sanofi and Regeneron’s Dupixent, among others.
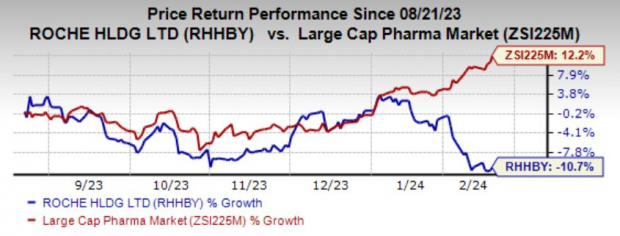
Market Performance and Outlook
Roche’s 2023 performance, excluding COVID-19-related sales, has been promising, particularly with the strong performance of drugs like Vabysmo, Ocrevus, Hemlibra, and Polivy. However, the outlook for 2024 seems to be less remarkable, as the company grapples with challenges such as competition from biosimilars for established drugs like Avastin, MabThera/Rituxan, and Herceptin. Additionally, Roche faces challenges from the approval of a higher dose of Eylea, a competitive drug to Vabysmo. These externalities might affect the company’s growth trajectory
Zacks Rank and Alternative Stock
Currently, Roche holds a Zacks Rank #4 (Sell). For investors seeking other options, Puma Biotechnology, Inc. has been identified as a Zacks Rank #1 (Strong Buy), attributing to its strong performance and potential growth in the market.
To conclude, the FDA’s approval of Xolair for food allergies represents a momentous achievement for Roche, with significant implications for the market. This development underscores the company’s commitment to addressing unmet medical needs and offers a glimmer of hope to allergy sufferers, potentially reshaping the landscape of food allergy treatments.