FDA’s Label Update for Dupixent
Regeneron Pharmaceuticals, Inc. (REGN) and its collaborator Sanofi (SNY) have recently announced a significant milestone. The FDA has given the green light to an updated label for Dupixent (dupilumab), expanding its indication for atopic dermatitis.
LIBERTY-AD-HAFT Study Data
The label update is a result of the late-stage LIBERTY-AD-HAFT study, which examined the efficacy and safety of Dupixent in 133 adult and adolescent patients with atopic dermatitis and moderate-to-severe hand and/or foot involvement.
According to the study, 40% of patients treated with Dupixent achieved clear or almost clear skin on hands and feet compared to 17% with a placebo. Additionally, 52% of Dupixent-treated patients saw a clinically meaningful reduction in itch on hands and feet compared to 14% with a placebo.
Dupixent’s Global Impact and Sales
Dupixent has garnered regulatory approvals worldwide for use in various conditions. The drug reported sales of $8.4 billion in the first nine months of 2023, reflecting a robust 34% increase. The label expansion will only serve to further propel its sales trajectory.
Challenges and Opportunities
While Dupixent continues to shine, Regeneron is facing challenges with its lead drug, Eylea. The latter suffered a setback as it reported disappointing fourth-quarter preliminary sales, with heightened competition impacting its performance.
However, the label update for Dupixent comes as a silver lining, bolstering the company’s prospects and reinforcing its foothold in the lucrative pharmaceutical space. Sanofi also expects significant growth in Dupixent sales, with potential opportunities in the chronic obstructive pulmonary disease market.
Positive Market Response and Competition
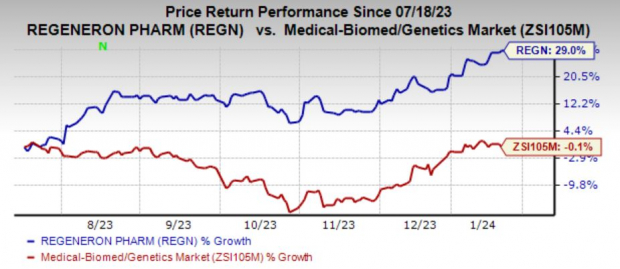
Regeneron’s shares have experienced a robust 29% surge in the past six months, indicating positive market sentiment. However, the industry at large has not fared as well, facing a minute 0.1% decline.
Moreover, Regeneron’s rival, Roche’s Vabysmo, has posed stiff competition, impacting Eylea sales. The uptake of Vabysmo has been exceptional, exerting pressure on Regeneron’s sales in the market.
Future Outlook and Top-Ranked Stocks
Despite these challenges, Regeneron remains optimistic about its future, particularly in solidifying its position in the lucrative oncology sector. The company has also been ranked as a Zacks Rank #1 (Strong Buy).
Another biotech stock, Puma Biotechnology, Inc. (PBYI), shares the same prestigious rank as Regeneron. Puma Biotechnology has shown promise with an upward trend in its earnings per share estimates and a substantial rise in its stock value over the past year.
Expanding Opportunities
Sanofi remains optimistic about the potential label expansion for Dupixent and its anticipated annual double-digit sales growth. The company is looking towards a lucrative future with vast opportunities in the chronic obstructive pulmonary disease space.
Conclusion
The revamped label for Dupixent marks a significant milestone for both Regeneron and Sanofi. It not only signifies a leap forward in the treatment of atopic dermatitis but also spells out potential market expansion and robust growth prospects for both companies. While facing challenges in some quarters, the prevalence of resilient and promising developments like Dupixent’s label update sets the stage for a compelling future in the pharmaceutical landscape.
5 Stocks Our Experts Predict Could Double In the Next Year
By submitting your email, you'll also get a free pivot & flow membership. A free daily market overview. You can unsubscribe at any time.