Viking Therapeutics, the brainchild behind VK2735, soared by 16.8% on Mar 26 following the release of favorable data from an early-phase trial on its oral obesity drug candidate. The experimental drug, an oral formula of VK2735, was examined in a phase I clinical study which demonstrated notable reductions in average body weight from baseline after 28 days of daily administration. Patients treated with the highest dose of VK2735, standing at 40mg, experienced an impressive 5.3% reduction in body weight, surpassing the 2.1% recorded in the placebo group.
The drug reported a remarkable mean body weight reduction of up to 3.3% when adjusted against placebo rates. At a 20mg dose, patients exhibited a weight loss of 1.1%. Furthermore, 57% of participants under the 40mg regimen achieved a weight loss of at least 5%. The drug displayed excellent tolerability across all doses, with no reported serious adverse effects.
Initial analysis by Viking suggests that extending the treatment period beyond the current 28-day mark may lead to further reductions in body weight. Patients who received higher doses appeared to sustain or even augment their weight loss by Day 34, six days post their last dose, registering weight losses of up to 3.6% relative to placebo. These results surging Wall Street’s expectations for weight loss, culminated in a sharp spike in Viking’s share price. Additionally, Viking disclosed its intent to propel VK2375 into mid-stage development by the culmination of the calendar year.
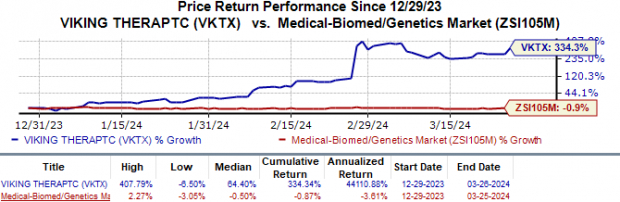
Year-to-date, shares of Viking Therapeutics have ascended by a remarkable 334.3%, starkly contrasting the industry’s 0.9% downturn.
Competitive Landscape in the Obesity Drug Market
Well before Viking Therapeutics’ jubilant ascent, the obesity drug landscape had piqued interest with Novo Nordisk’s unveiling of Wegovy, securing FDA endorsement in 2021 for chronic weight management in overweight or obese adults. Since its approval, demand for Novo Nordisk’s product has been on an upswing.
Recently, Eli Lilly debuted Zepbound, its cutting-edge obesity drug, backed by FDA approval in December and a subsequent launch in January. Aiming to tap into a burgeoning market for obesity drugs, Lilly has been exploring multiple other obesity candidates within its pipeline, including retatrutide and orforglipron. The acquisition of private biotech firm Versanis last year further fortified Lilly’s stance in the obesity arena.
In the pursuit of innovation within the obesity sector, AstraZeneca and Roche have also joined the fray. Roche’s acquisition of privately held Carmot Therapeutics for $2.7 billion in December marked its foray into the obesity sphere, granting access to Carmot’s unique incretin portfolio. On the other hand, AstraZeneca secured exclusive global rights from Eccogene to develop ECC5004, an oral drug targeted at obesity, type-II diabetes, and other cardiometabolic conditions, reflecting a concerted effort to expand its foothold in this competitive landscape.
Viking Therapeutics, Inc. Price
Viking Therapeutics, Inc. price | Viking Therapeutics, Inc. Quote
Zacks Rank
Viking currently holds a Zacks Rank #3 (Hold). For the complete list of Zacks Rank #1 (Strong Buy) stocks, click here.
7 Best Stocks for the Next 30 Days
Just released: Experts highlight 7 elite stocks from the current selection of 220 Zacks Rank #1 Strong Buys. These stocks are perceived to be “Most Likely for Early Price Pops.”
Interested in the latest recommendations from Zacks Investment Research? Download 7 Best Stocks for the Next 30 Days for free! Click here.
Read the original article on Zacks.com here.
The author’s views are personal and do not necessarily reflect those of Nasdaq, Inc.